Chapter 22: Q18. (page 892)
Draw a stepwise mechanism for the following reaction.
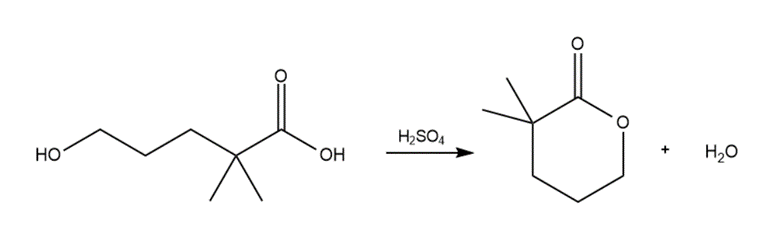
Short Answer
Answer
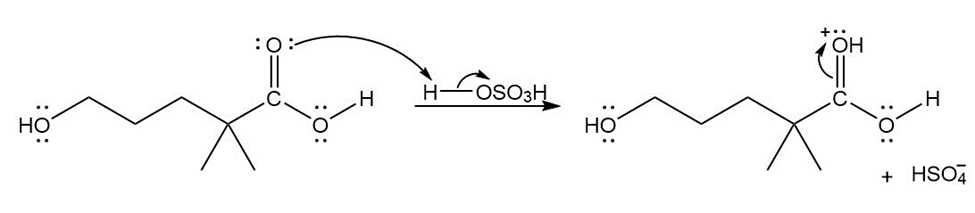
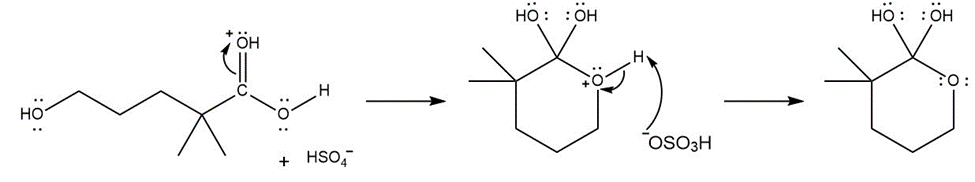

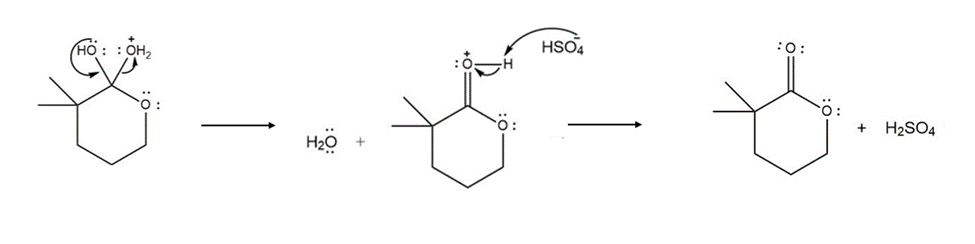
Formation of the final product
Learning Materials
Features
Discover
Chapter 22: Q18. (page 892)
Draw a stepwise mechanism for the following reaction.

Answer




Formation of the final product
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:What reagents are needed to convert phenylacetonitrile () to each compound:
Draw all stereoisomers formed in each reaction.
What organocuprate reagent is needed to convert CH3CH2COCIto each ketone?
a.
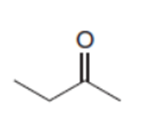
b.

c.
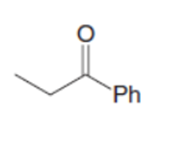
Identify the lettered compounds in the following reaction scheme. Compounds F, G, and K are isomers of molecular formula C13H18O. How could 1HNMR spectroscopy distinguish these three compounds from each other?

Question: Give the IUPAC or common name for each compound.
a. 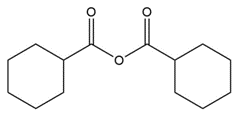
b. 
c. 
d. 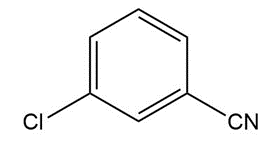
e. 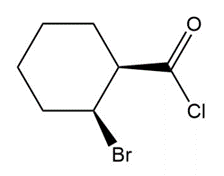
f.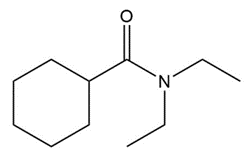
What do you think about this solution?
We value your feedback to improve our textbook solutions.