Chapter 22: Q19. (page 894)
Question: What product is formed when acetic acid is treated with each reagent: (a) ; (b) , then heat; (c) + DCC?
Short Answer
Answer
a.
b. 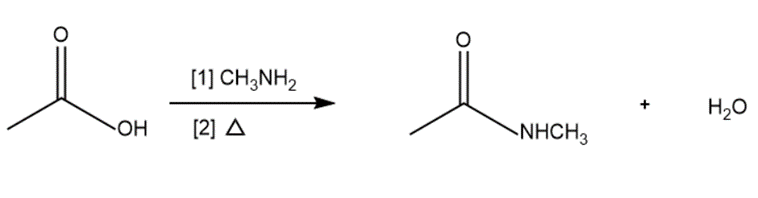
c.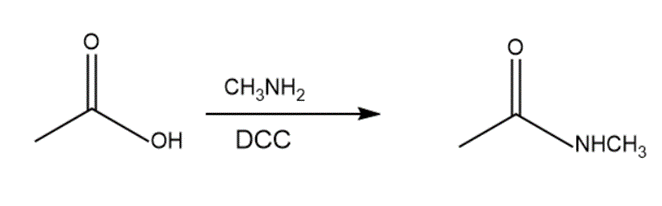
Learning Materials
Features
Discover
Chapter 22: Q19. (page 894)
Question: What product is formed when acetic acid is treated with each reagent: (a) ; (b) , then heat; (c) + DCC?
Answer
a.
b. 
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
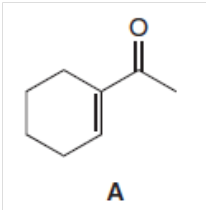
Draw the product formed when the -unsaturated ketone A is treated with each reagent.
(a) NaBH4, CH3OH
(b) H2 (1 equiv), Pd-C
(c) H2 (excess), Pd-C
(d) [1] CH3Li ; [2]H2O
(e) [1] CH3CH2MgBr ; [2]H2O
(f) [1] ; [2]H2O
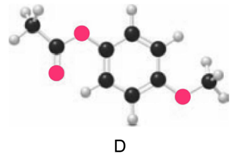
Question: Which ester, C or D, is more reactive in nucleophilic acyl substitution? Explain your reasoning.
Oct-1-yne (HCCCH2CH2CH2CH2CH2CH3) reacts rapidly with NaH, forming a gas that bubbles out of the reaction mixture, as one product. Oct-1-yne also reacts rapidly with CH3MgBr , and a different gas is produced. Write balanced equations for both reactions and identify the gases formed.
Draw the products formed (including stereoisomers) when each compound is reduced with NaBH4in CH3OH.
Using protecting groups, show how estrone can be converted to ethynylestradiol, a widely used oral contraceptive.
What do you think about this solution?
We value your feedback to improve our textbook solutions.