Chapter 22: 26P (page 868)
Using protecting groups, show how estrone can be converted to ethynylestradiol, a widely used oral contraceptive.
Short Answer
Answer
Estrone can be converted to ethynylestradiol by the following method:
Learning Materials
Features
Discover
Chapter 22: 26P (page 868)
Using protecting groups, show how estrone can be converted to ethynylestradiol, a widely used oral contraceptive.
Answer
Estrone can be converted to ethynylestradiol by the following method:
All the tools & learning materials you need for study success - in one app.
Get started for free
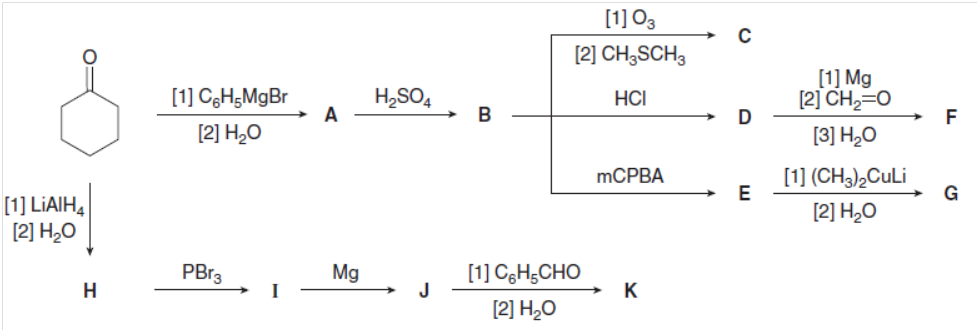
Identify the lettered compounds in the following reaction scheme. Compounds F, G, and K are isomers of molecular formula C13H18O. How could 1HNMR spectroscopy distinguish these three compounds from each other?
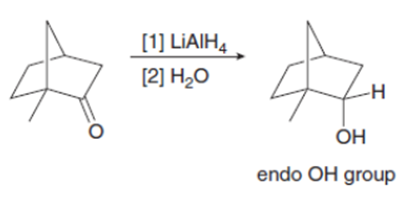
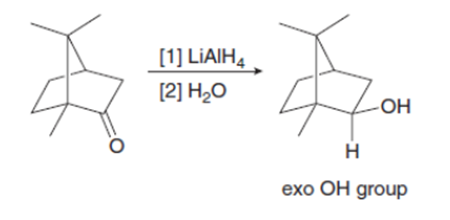
Explain why metal hydride reduction gives an endo alcohol as the major product in one reaction given below and an exo alcohol as the major product in the other reaction.
Linalool (the Chapter 9 opening molecule) and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can’t linalool beprepared by a similar pathway?
Question: Explain why is a stronger acid and a weaker base than .
Question: Draw the products of each reaction and indicate the stereochemistry at any stereogenic centers.
a.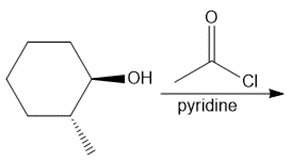
b. 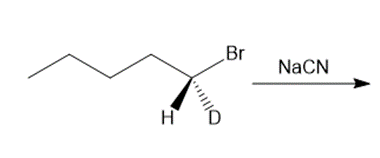
c.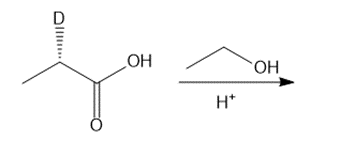
d. 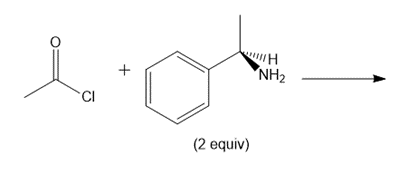
What do you think about this solution?
We value your feedback to improve our textbook solutions.