Chapter 22: Q51. (page 916)
Question: Draw the products of each reaction and indicate the stereochemistry at any stereogenic centers.
a.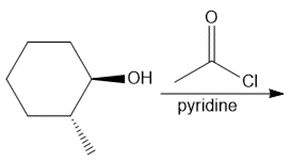
b. 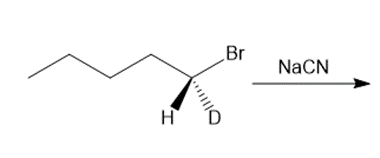
c.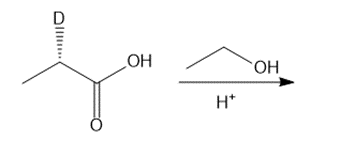
d. 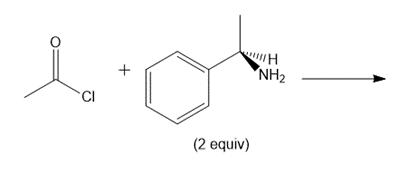
Short Answer
Answer
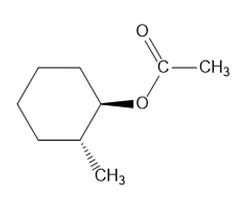
a.
b.
c.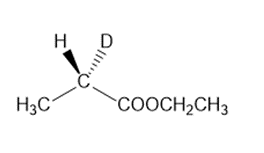
d.
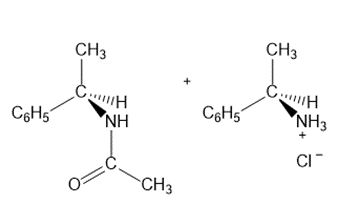
Learning Materials
Features
Discover
Chapter 22: Q51. (page 916)
Question: Draw the products of each reaction and indicate the stereochemistry at any stereogenic centers.
a.
b. 
c.
d. 
Answer

a.
b.
c.
d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for the following reduction.

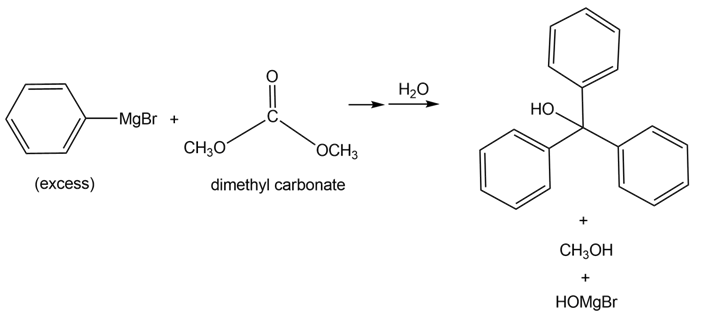
Tertiary alcohols can be formed by the reaction of dimethyl carbonate [(CH3O)2CO ] with excess Grignard reagent. Draw a stepwise mechanism for the following transformation.
Draw the products formed from LiAlH4 reduction of each compound.
Question:What reagents are needed to convert phenylacetonitrile () to each compound:
What Grignard reagent and aldehyde (or ketone) are needed to prepare each alcohol? Show all possible routes
(a)
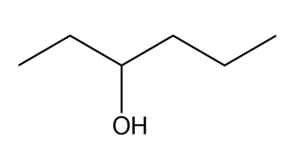
(b)
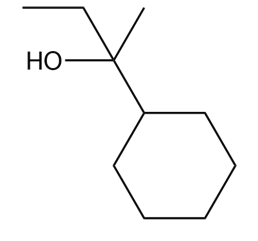
(c)
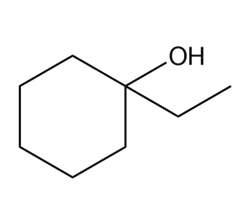
What do you think about this solution?
We value your feedback to improve our textbook solutions.