Chapter 22: Q43. (page 915)
Question: Explain why is a stronger acid and a weaker base than .
Short Answer
Answer
The given amide is a stronger acid and a weaker base than as the lone pair is not available for donation.
Learning Materials
Features
Discover
Chapter 22: Q43. (page 915)
Question: Explain why is a stronger acid and a weaker base than .
Answer
The given amide is a stronger acid and a weaker base than as the lone pair is not available for donation.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products formed when CH3COCH2CH2CH=CH2is treated with each reagent: (a)LiAlH4, then H2O ; (b) NaBH4in CH3OH ; (c) H2(1 equiv), Pd-C; (d)H2(excess), Pd-C; (e)NaBH4 (excess) inCH3OH ; (f) NaBD4in CH3OH
Draw a stepwise mechanism for the following reduction.
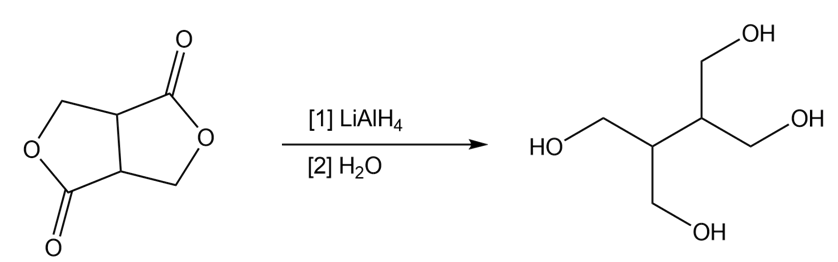
Write the step(s) needed to convert CH3CH2Brto each reagent: (a)CH3CH2Li (b) CH3CH2MgBr(c) (CH3CH2)2CuLi
Why can’t 1-methylcyclohexanol be prepared from a carbonyl compound by reduction?
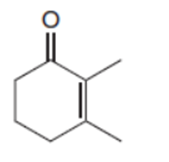
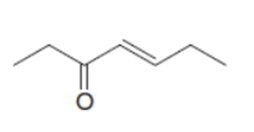
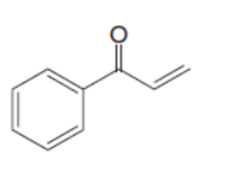
Draw the product when each compound is treated with either (CH3)2CuLi , followed by H2O , HCCLi , followed by H2O .
a.
b.
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.