Chapter 22: 6P (page 868)
Why can’t 1-methylcyclohexanol be prepared from a carbonyl compound by reduction?
Short Answer
Answer
In 1-methyl cyclohexanol, tertiary alcohol is present, hence it can’t be reduced from any carbonyl compound.
Learning Materials
Features
Discover
Chapter 22: 6P (page 868)
Why can’t 1-methylcyclohexanol be prepared from a carbonyl compound by reduction?
Answer
In 1-methyl cyclohexanol, tertiary alcohol is present, hence it can’t be reduced from any carbonyl compound.
All the tools & learning materials you need for study success - in one app.
Get started for free
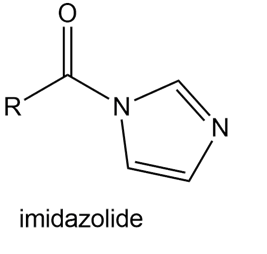
Question: Explain why imidazolides are much more reactive than other amides in nucleophilic acyl substitution.
Question:Outline two different ways that butan-2-one can be prepared from a nitrile and a Grignard reagent.
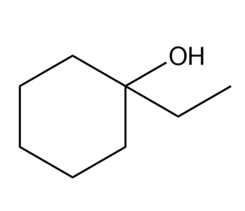
What Grignard reagent and aldehyde (or ketone) are needed to prepare each alcohol? Show all possible routes
(a)
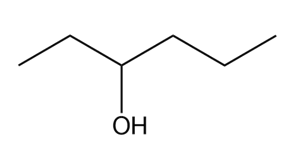
(b)

(c)
Question: Explain why is a stronger acid and a weaker base than .
Question: Give the structure corresponding to each name.
What do you think about this solution?
We value your feedback to improve our textbook solutions.