Chapter 22: Q16. (page 892)
Question: Draw the products of each reaction.
a.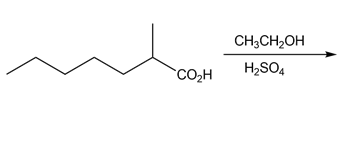
b. 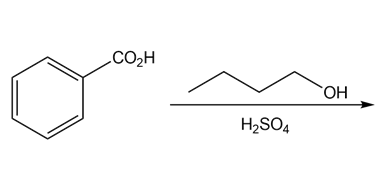
c.
d. 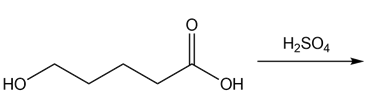
Short Answer
Answer
a. 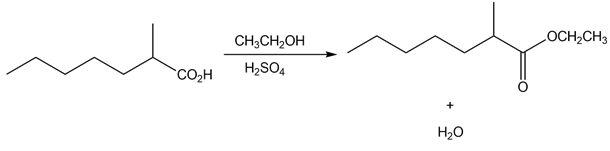
b.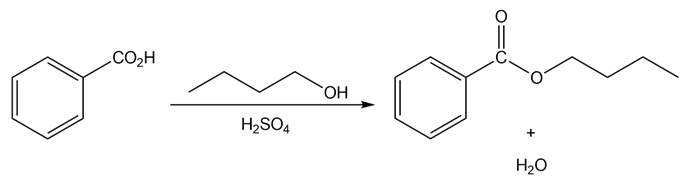
c. 

d.
Learning Materials
Features
Discover
Chapter 22: Q16. (page 892)
Question: Draw the products of each reaction.
a.
b. 
c.
d. 
Answer
a. 
b.
c. 

d.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the three possible resonance structures for an acid bromide, . Then, using the pKa values in Appendix A, decide if RCOBr is more or less stabilized by resonance than a carboxylic acid (RCOOH).
Question: Explain why is a stronger acid and a weaker base than .
Question: (a) Give an acceptable name for each compound. (b) Draw the organic products formed when A or B is treated with each reagent: [1](excess), then ; [4] , then .

Question: Draw a stepwise mechanism for the formation of A from an alcohol and acid chloride. A was converted in one step to Blattella quinone, the sex pheromone of the female German cockroach Blattella germanica.

Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs.
What do you think about this solution?
We value your feedback to improve our textbook solutions.