Chapter 22: Q15. (page 890)
Question: Draw the products of each reaction
a.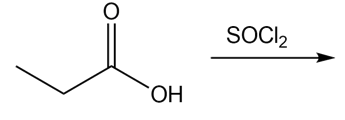
b. 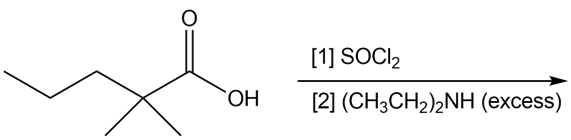
Short Answer
Answer
Product formed:
(a)
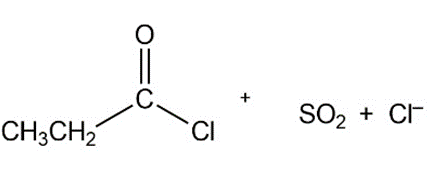
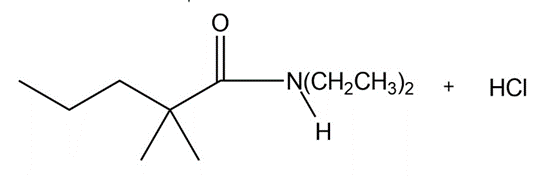
b.
Learning Materials
Features
Discover
Chapter 22: Q15. (page 890)
Question: Draw the products of each reaction
a.
b. 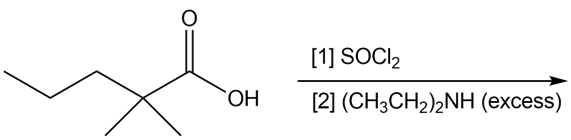
Answer
Product formed:
(a)

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What two monomers are needed to prepare nylon 6,10?
Oct-1-yne (HCCCH2CH2CH2CH2CH2CH3) reacts rapidly with NaH, forming a gas that bubbles out of the reaction mixture, as one product. Oct-1-yne also reacts rapidly with CH3MgBr , and a different gas is produced. Write balanced equations for both reactions and identify the gases formed.
Draw the products of the following reactions with organometallic reagents.
a.
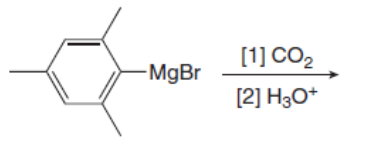
b.
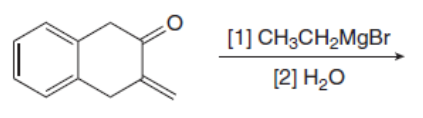
c.
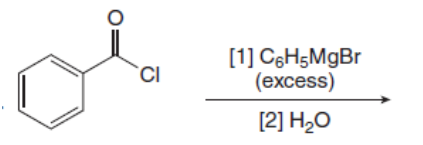
d.
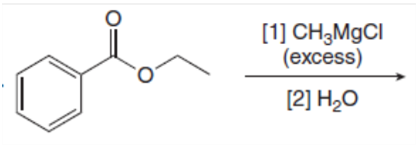
e.
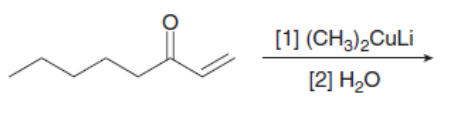
f.
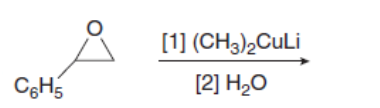
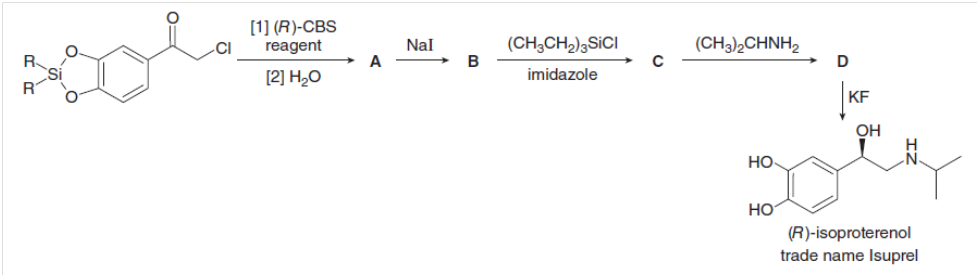
Fill in the lettered products (A–D) in the synthesis of (R)-isoproterenol, a drug that increases heart rate and dilates lung passages.
Draw the structure of both an acid chloride and an ester that can be used to prepare each compound by reduction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.