Chapter 22: 31P (page 868)
Draw two different ways to prepare each ketone from an acid chloride and an organocuprate reagent.
a.
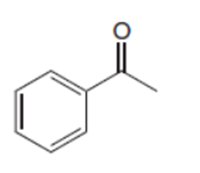
b.
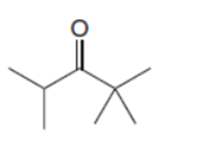
Short Answer
Answer
The compounds used to prepare the given compounds are:
a.
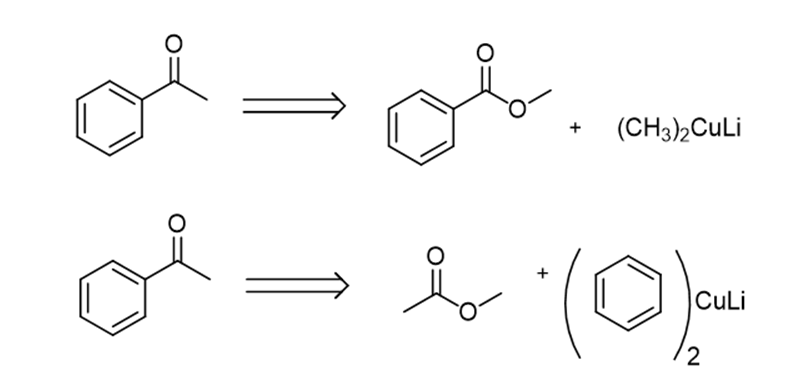
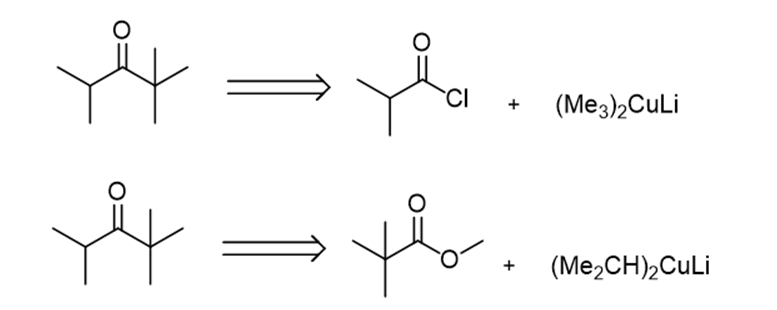
b.
Learning Materials
Features
Discover
Chapter 22: 31P (page 868)
Draw two different ways to prepare each ketone from an acid chloride and an organocuprate reagent.
a.

b.

Answer
The compounds used to prepare the given compounds are:
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw a stepwise mechanism for the formation of A from an alcohol and acid chloride. A was converted in one step to Blattella quinone, the sex pheromone of the female German cockroach Blattella germanica.
Question: Draw the structure of Kodel, a polyester formed from 1,4-dihydroxymethylcyclohexane and terephthalic acid. Explain why fabrics made from Kodel are stiff and crease-resistant.
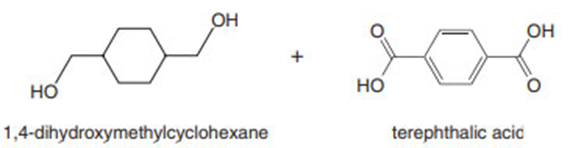
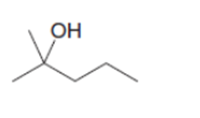
What epoxide is needed to convertCH3CH2MgBrto each of the following alcohols, after quenching with water?
a.
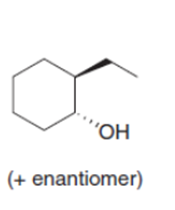
b.
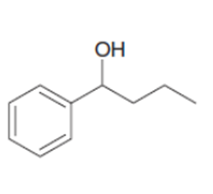
c.
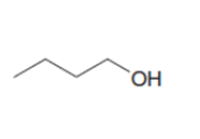
d.
Question: Rank the compounds in each group in order of increasing reactivity in nucleophilic acyl substitution.
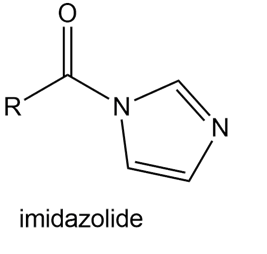
Question: Explain why imidazolides are much more reactive than other amides in nucleophilic acyl substitution.
What do you think about this solution?
We value your feedback to improve our textbook solutions.