Chapter 22: Q10. (page 884)
Question: Rank the compounds in each group in order of increasing reactivity in nucleophilic acyl substitution.
Short Answer
Answer
a.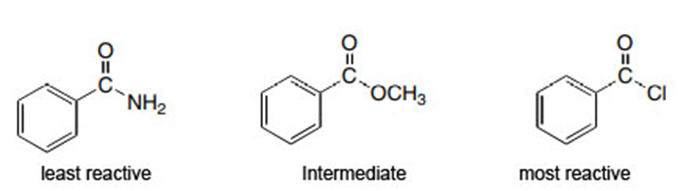
b.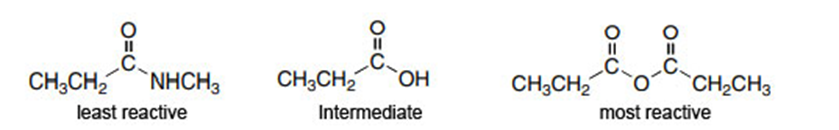
Learning Materials
Features
Discover
Chapter 22: Q10. (page 884)
Question: Rank the compounds in each group in order of increasing reactivity in nucleophilic acyl substitution.
Answer
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the products of each reaction.
a.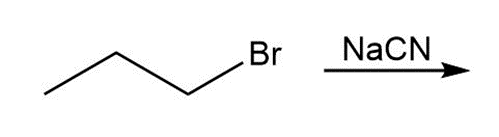
b. 
c. 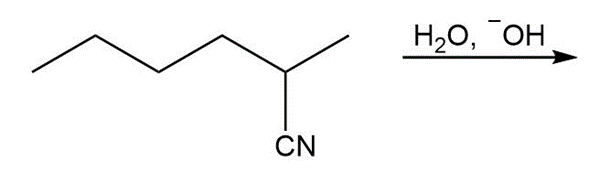
Question: (a) Give an acceptable name for each compound. (b) Draw the organic products formed when A or B is treated with each reagent: [1](excess), then ; [4] , then .

Question: What product is formed when acetic acid is treated with each reagent: (a) ; (b) , then heat; (c) + DCC?
Question: Explain why imidazolides are much more reactive than other amides in nucleophilic acyl substitution.

Using protecting groups, show how estrone can be converted to ethynylestradiol, a widely used oral contraceptive.
What do you think about this solution?
We value your feedback to improve our textbook solutions.