Chapter 22: Q85. (page 923)
Question: Draw a stepwise mechanism for the following reaction, a key step in the synthesis of linezolid, an antibacterial agent.
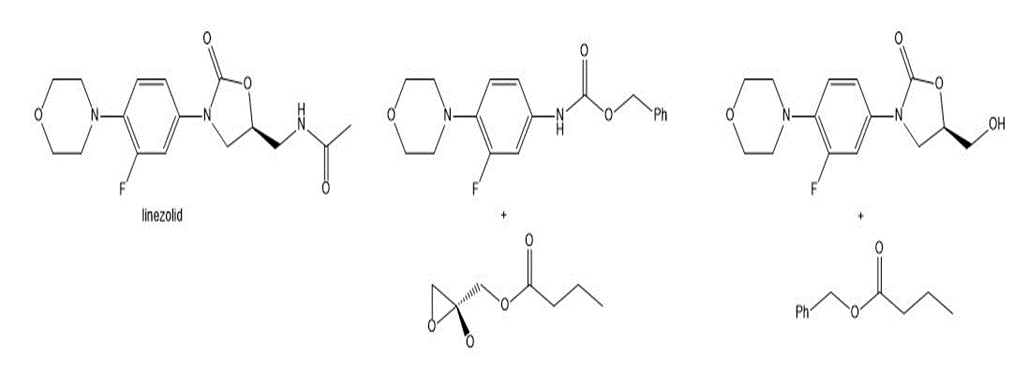
Short Answer
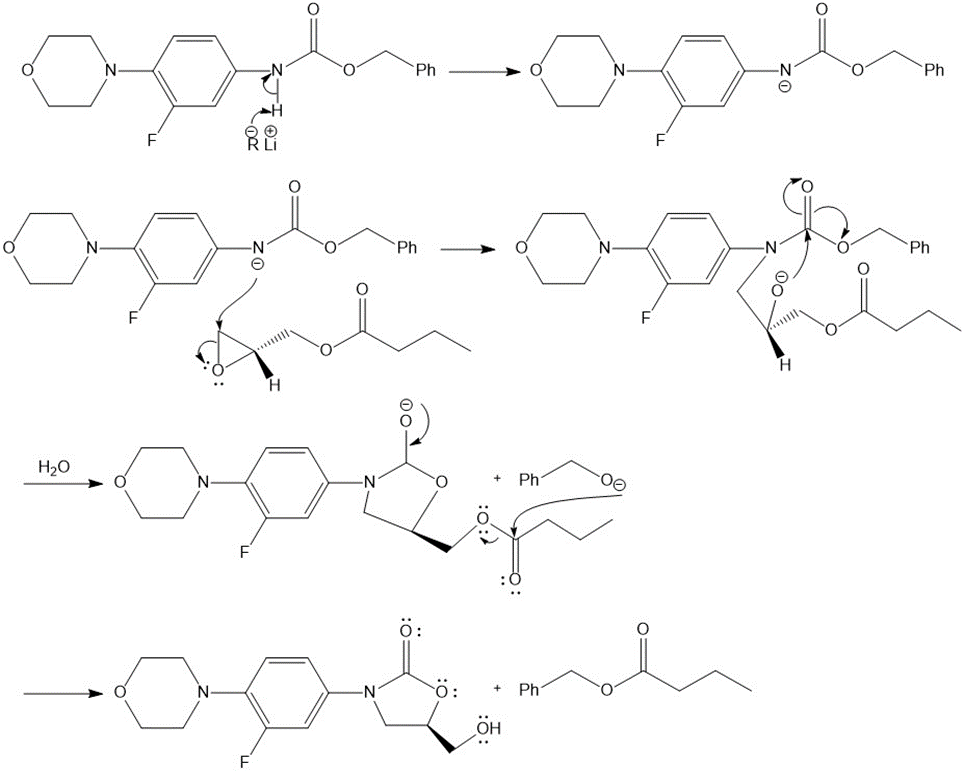
Answer
Learning Materials
Features
Discover
Chapter 22: Q85. (page 923)
Question: Draw a stepwise mechanism for the following reaction, a key step in the synthesis of linezolid, an antibacterial agent.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Which compound in each pair is more reactive towards nucleophilic attack?
What Grignard reagent and carbonyl compound are needed to prepare each alcohol? As shown in part (d), 3o alcohols with three different R groups on the carbon bonded to the OH group can be prepared by three different Grignard reactions.
Question: How do the following experimental results support the resonance description of the relative stability of acid chlorides compared to amides? The C-Cl bond lengths in and CH3COCl are identical (178 pm), but the C-N bond in is shorter than the C-N bond in (135 pm versus 147 pm).
Draw the product formed when each organometallic reagent is treated with H2O.
Draw the products (including stereochemistry) of the following reactions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.