Chapter 22: 20P (page 868)
Draw the product formed when each organometallic reagent is treated with H2O.
Short Answer
Answer
The products formed when each organometallic reagent is treated with water are shown below:
Learning Materials
Features
Discover
Chapter 22: 20P (page 868)
Draw the product formed when each organometallic reagent is treated with H2O.
Answer
The products formed when each organometallic reagent is treated with water are shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
stereochemistry of the products of reduction depends on the reagent used,as you learned in Sections 20.5 and 20.6. With this in mind, how would you convert3,3-dimethylbutan-2-one [CH3COC(CH3)3] to: (a) racemic 3,3-dimethylbutan-2-ol[CH3CH(OH)C (CH3)3] ;(b) only (R)-3,3-dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?
Draw a stepwise mechanism for the following reaction.
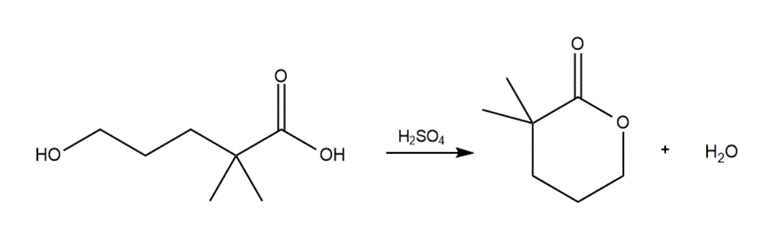
Question:(a) Propose an explanation for the difference in the frequency of the carbonyl absorptions of phenyl acetate (1765 ) and cyclohexyl acetate (1738 ).(b) Which carbonyl group is more effectively stabilized by resonance?(c) Which ester reacts faster when treated with aqueous base?
Draw the products formed (including stereoisomers) when each compound is reduced with NaBH4in CH3OH.
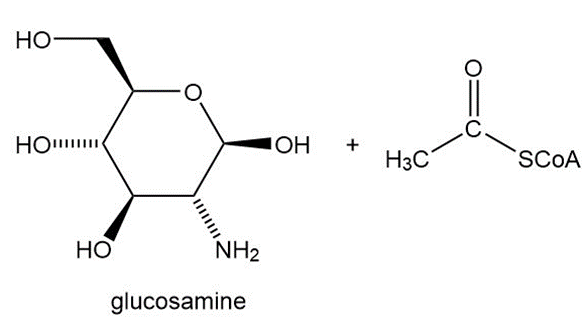
Question: Glucosamine is a dietary supplement available in many over-the-counter treatments for osteoarthritis. Reaction of acetyl CoA with glucosamine forms NAG, N-acetylglucosamine, the monomer used to form chitin, the carbohydrate that forms the rigid shells of lobsters and crabs. What is the structure of NAG?
What do you think about this solution?
We value your feedback to improve our textbook solutions.