Chapter 24: Q.45. (page 962)
Question: Draw the product of each Robinson annulation from the given starting materials using in solution.
Short Answer
Answer
a.
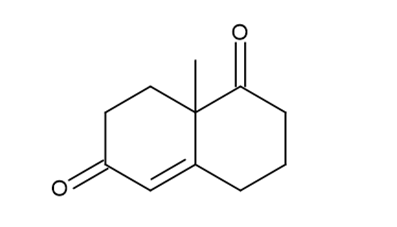
b.
Learning Materials
Features
Discover
Chapter 24: Q.45. (page 962)
Question: Draw the product of each Robinson annulation from the given starting materials using in solution.
Answer
a.

b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which carbonyl compounds do not undergo an aldol reaction when treated with OH- in H2O ?
Question: Devise a stepwise mechanism for the following reaction. (Hint: The mechanism begins with the conjugate addition of .)
Question: Draw the products formed in the crossed aldol reaction of phenylacetaldehyde with each compound: (a) ; (b) ; (c) .
Question: Acid-catalyzed dehydration of β-hydroxy carbonyl compounds occurs by the mechanism discussed in Section 9.8. With this in mind, draw a stepwise mechanism for the following reaction.

Question: What two β-keto esters are formed in the Dieckmann reaction of the following diester?

What do you think about this solution?
We value your feedback to improve our textbook solutions.