Chapter 24: Q.69. (page 962)
Question: Devise a stepwise mechanism for the following reaction. (Hint: The mechanism begins with the conjugate addition of .)
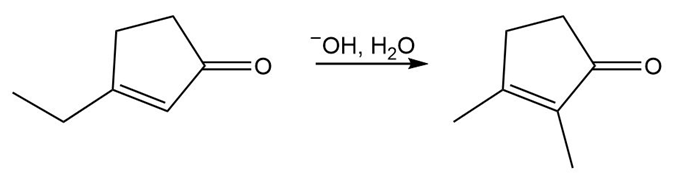
Short Answer
Answer
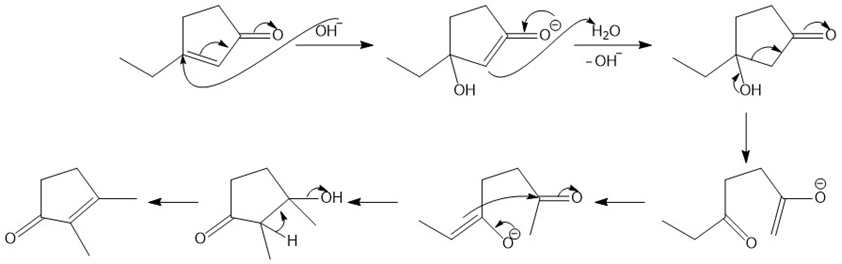
Mechanism of rearrangement reaction
Learning Materials
Features
Discover
Chapter 24: Q.69. (page 962)
Question: Devise a stepwise mechanism for the following reaction. (Hint: The mechanism begins with the conjugate addition of .)

Answer

Mechanism of rearrangement reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Draw the product formed from a Michael reaction with the given starting materials using.
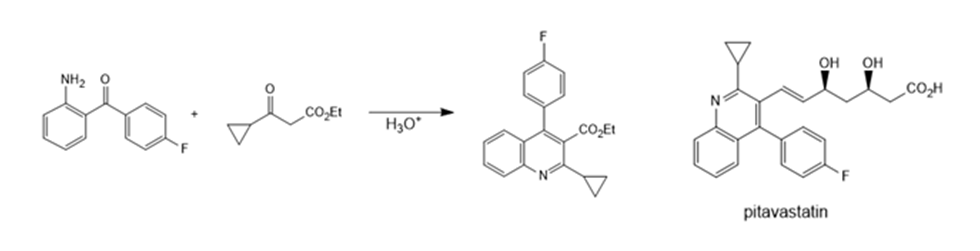
Question: Draw a stepwise mechanism for the following reaction, one step in the synthesis of the cholesterol-lowering drug pitavastatin, marketed in Japan as a calcium salt under the name Livalo.
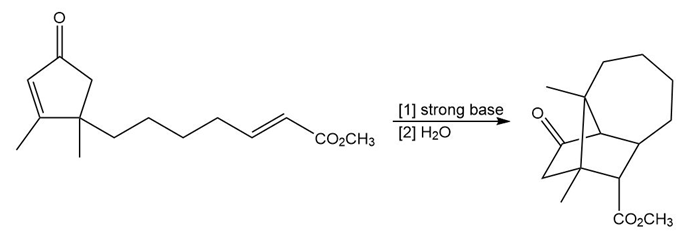
Question: Draw a stepwise mechanism for the following reaction. (Hint: Two Michael reactions are needed.)
Question: What starting materials are needed to synthesize each compound by a Robinson annulation?
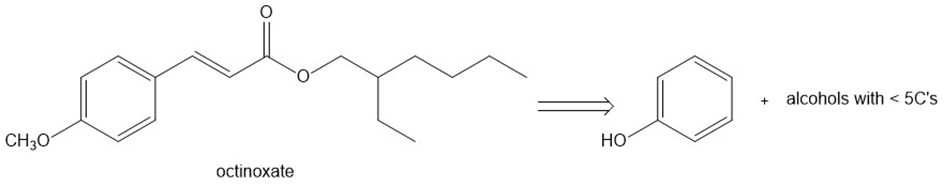
Question: Octinoxate is an unsaturated ester used as an active ingredient in sunscreens.(a) What carbonyl compounds are needed to synthesize this compound using a condensation reaction?(b) Devise a synthesis of octinoxate from the given organic starting materials and any other needed reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.