Chapter 24: Q.7. (page 962)
Question: Draw the products formed in each crossed aldol reaction.
a.
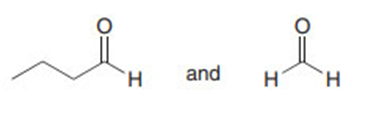
b.
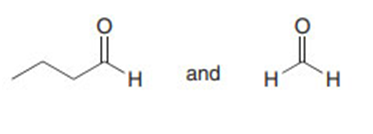
Short Answer
Answer
a.
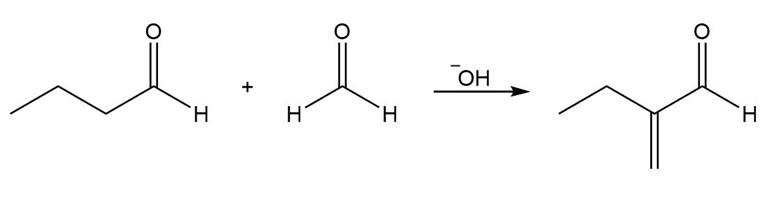
b.
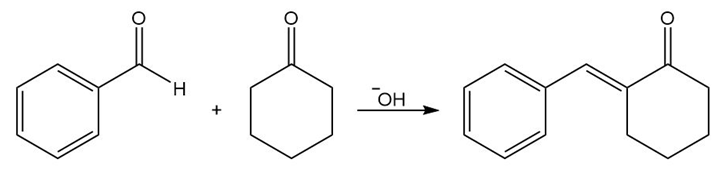
Learning Materials
Features
Discover
Chapter 24: Q.7. (page 962)
Question: Draw the products formed in each crossed aldol reaction.
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
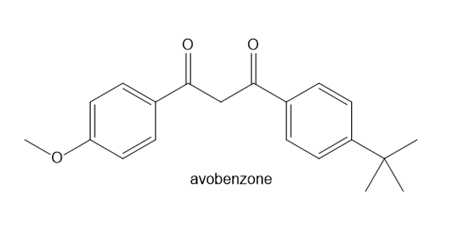
Question: Avobenzone is a conjugated compound that absorbs ultraviolet light with wavelengths in the 320–400 nm region, so it is a common ingredient in commercial sunscreens. Write out two different crossed Claisen reactions that form avobenzone.
Question: Which carbonyl compounds do not undergo an aldol reaction when treated with OH- in H2O ?
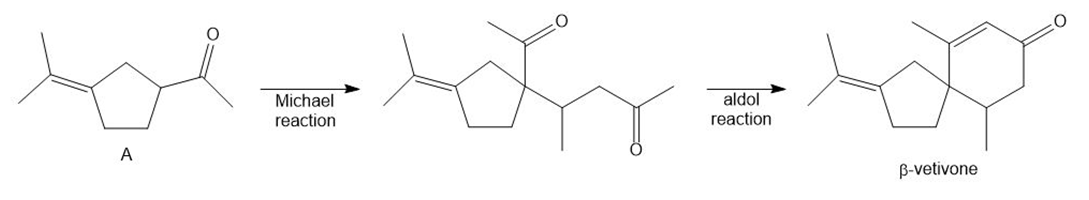
Question: Which of the following compounds can serve as Michael acceptors?
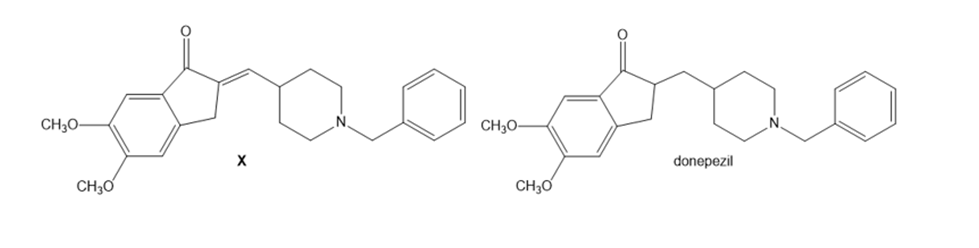
Question: A key step in the synthesis of donepezil (trade name Aricept) is a directed aldol reaction that forms -unsaturated carbonyl compound X. What carbonyl starting materials are needed to prepare X using a directed aldol reaction? What reagents are needed to convert X to donepezil?
What do you think about this solution?
We value your feedback to improve our textbook solutions.