Chapter 24: Q.2. (page 962)
Question: Which carbonyl compounds do not undergo an aldol reaction when treated with OH- in H2O ?
Short Answer
Answer
- No
- Yes
- No
- Yes
Learning Materials
Features
Discover
Chapter 24: Q.2. (page 962)
Question: Which carbonyl compounds do not undergo an aldol reaction when treated with OH- in H2O ?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
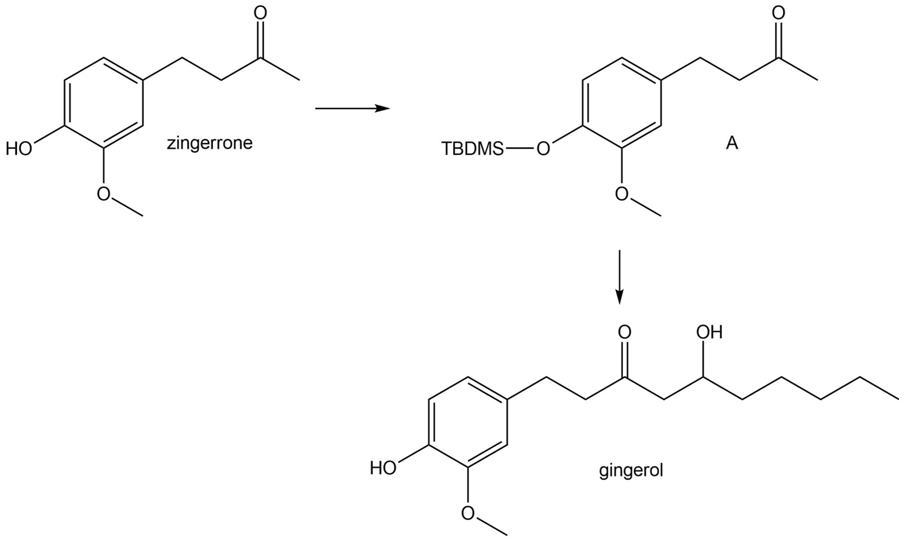
Question: Zingerone, a spicy-sweet component of cooked ginger, can be converted to its protected TBDMS ether A, as we learned in Chapter 20. How can A be converted to gingerol, a compound present in fresh ginger, using a directed aldol reaction as a key step?
Question: Draw the product formed when each dicarbonyl compound undergoes an intramolecular aldol reaction followed by dehydration.
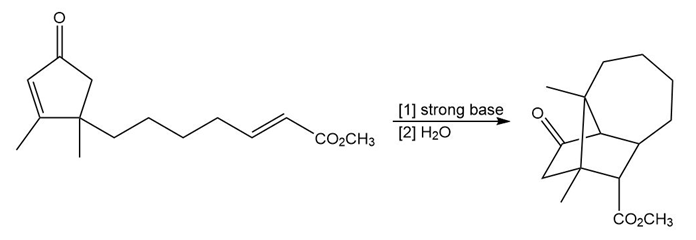
Question: Draw a stepwise mechanism for the following reaction. (Hint: Two Michael reactions are needed.)
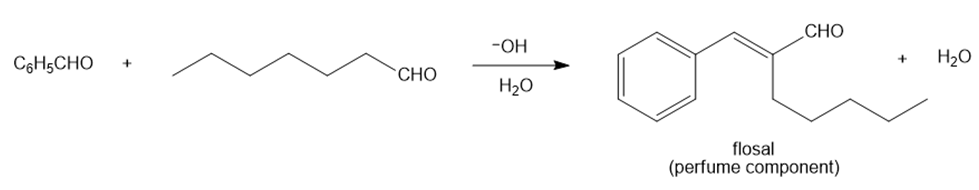
Question: 2-Pentylcinnamaldehyde, commonly called flosal, is a perfume ingredient with a jasmine-like odor. Flosal is an -unsaturated aldehyde made by a crossed aldol reaction between benzaldehyde and heptanal , followed by dehydration. Draw a stepwise mechanism for the following reaction that prepares flosal.
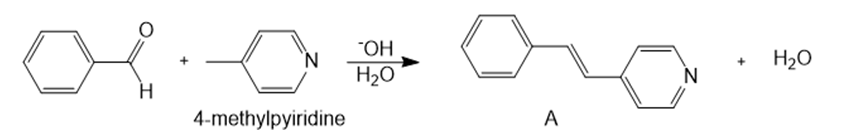
Question: 4-Methylpyridine reacts with benzaldehyde in the presence of a base to form A.
(a) Draw a stepwise mechanism for this reaction.
(b) Would you expect 2-methylpyridine or 3-methylpyridine to undergo a similar type of condensation reaction? Explain why or why not.
What do you think about this solution?
We value your feedback to improve our textbook solutions.