Chapter 24: Q.32. (page 962)
Question: Draw the product formed when each dicarbonyl compound undergoes an intramolecular aldol reaction followed by dehydration.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 24: Q.32. (page 962)
Question: Draw the product formed when each dicarbonyl compound undergoes an intramolecular aldol reaction followed by dehydration.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Draw the organic products formed in each reaction.
Question: What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous ?
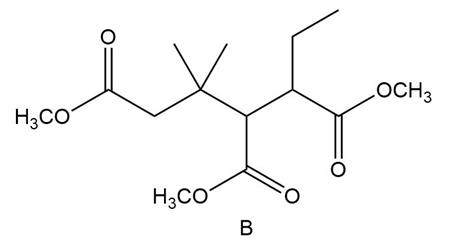
Question:Even though B contains three ester groups, a single Dieckmann product results when B is treated with in , followed by . Draw the structure and explain why it is the only product formed.
Question:Draw the product formed from a Claisen reaction with the given starting materials using –OEt, EtOH.
Question: What starting materials are needed to synthesize each compound using an aldol or similar reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.