Chapter 24: Q.44. (page 962)
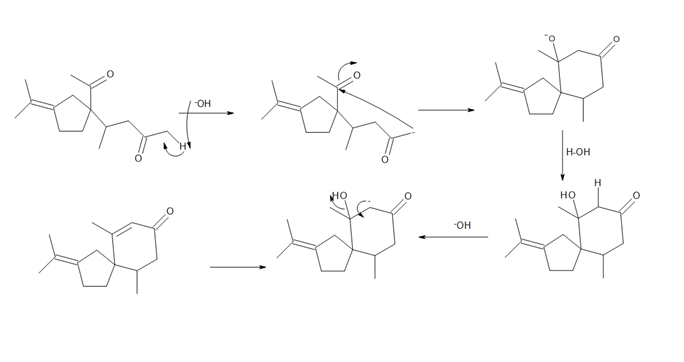
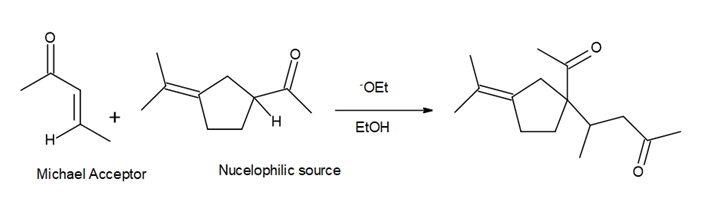
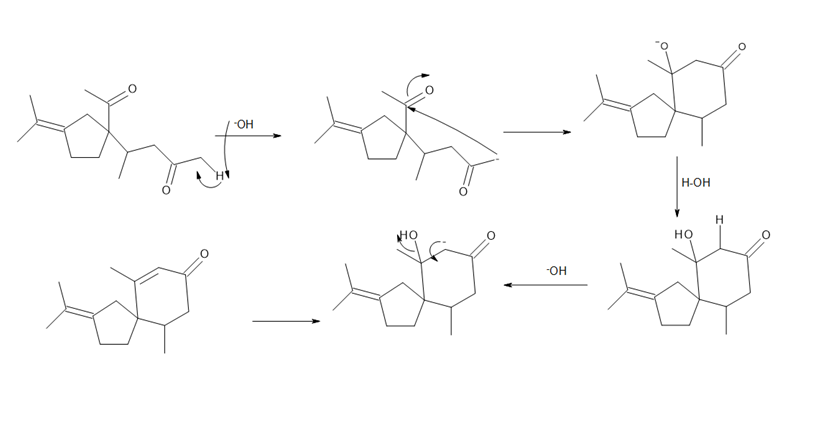
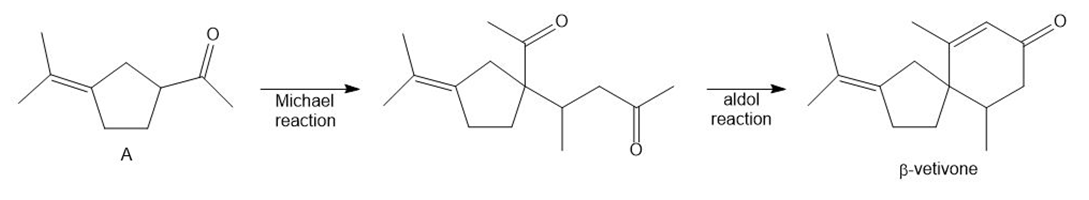
- Question:β -Vetivone is isolated from vetiver, a perennial grass that yields a variety of compounds usedin traditional eastern medicine, pest control, and fragrance. In one synthesis, ketone A is converted to β-vetivone by a two-step process: Michael reaction, followed by intramolecular 00a00000ldol reaction.000 (a) What Michael acceptor is needed for the conjugate addition? (See Problem23.61 for another method to form the bicyclic ring system of β-vetivone.) (b) Draw a stepwise mechanism for the aldol reaction, which forms the six-membered ring.
Short Answer
Answer
a. Michael acceptor:
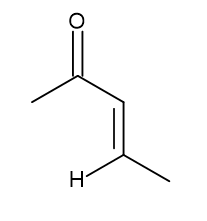
b.