Chapter 24: Q.8. (page 962)
Question: Draw the products formed in the crossed aldol reaction of phenylacetaldehyde with each compound: (a) ; (b) ; (c) .
Short Answer
Answer
a.
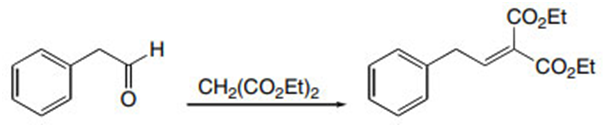
b.
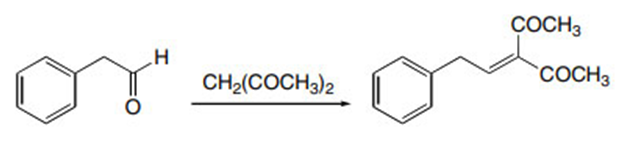
c.
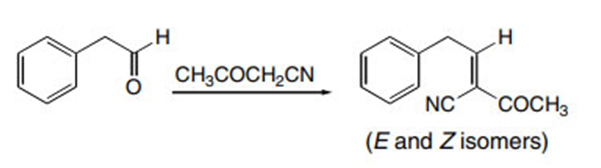
Learning Materials
Features
Discover
Chapter 24: Q.8. (page 962)
Question: Draw the products formed in the crossed aldol reaction of phenylacetaldehyde with each compound: (a) ; (b) ; (c) .
Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
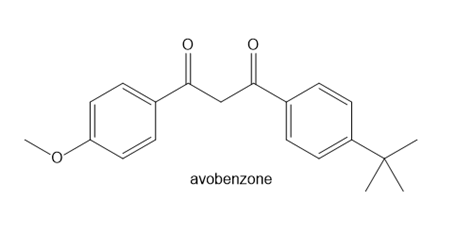
Question: Avobenzone is a conjugated compound that absorbs ultraviolet light with wavelengths in the 320–400 nm region, so it is a common ingredient in commercial sunscreens. Write out two different crossed Claisen reactions that form avobenzone.
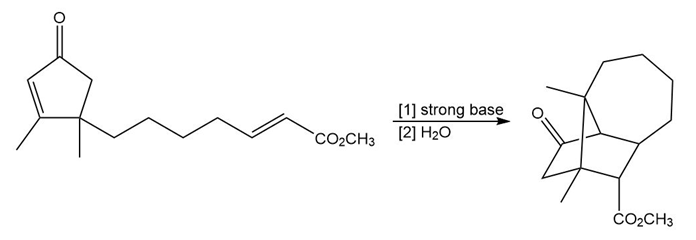
Question: Draw a stepwise mechanism for the following reaction. (Hint: Two Michael reactions are needed.)
Question: Draw the products formed in each crossed aldol reaction.
a.
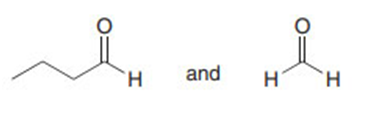
b.
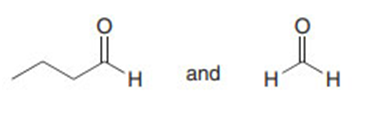
Question: What aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the following compounds?
Question: What dicarbonyl compound is needed to prepare each compound by an intramolecular aldol reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.