Chapter 24: Q.35. (page 962)
Question: What dicarbonyl compound is needed to prepare each compound by an intramolecular aldol reaction?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 24: Q.35. (page 962)
Question: What dicarbonyl compound is needed to prepare each compound by an intramolecular aldol reaction?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the aldol product formed from each pair of starting materials using .
Question:Draw the product formed from a Michael reaction with the given starting materials using.
Question: Draw a stepwise mechanism for the following reaction. (Hint: Two Michael reactions are needed.)
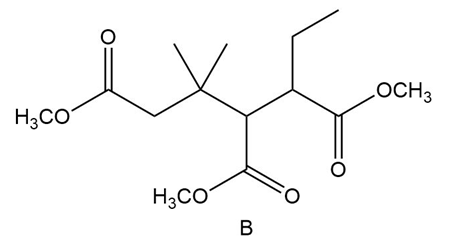
Question:Even though B contains three ester groups, a single Dieckmann product results when B is treated with in , followed by . Draw the structure and explain why it is the only product formed.
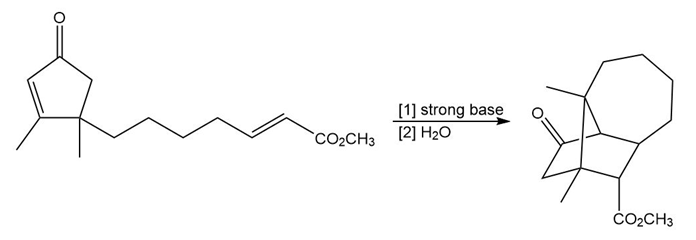
Question: Propose a stepwise mechanism for the following reaction of a -keto ester. Suggest a reason why this rearrangement reaction occurs.
What do you think about this solution?
We value your feedback to improve our textbook solutions.