Chapter 17: Q.20. (page 641)
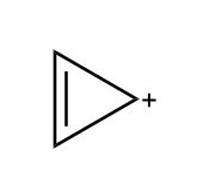
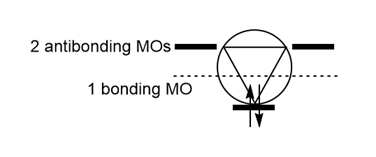
Question: Use the inscribed polygon method to show why the following cation is aromatic.
Short Answer
Expert verified
Answer
Learning Materials
Features
Discover
Chapter 17: Q.20. (page 641)
Question: Use the inscribed polygon method to show why the following cation is aromatic.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which compound in each pair is the stronger acid?
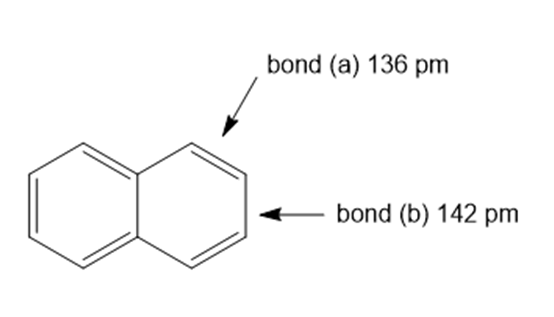
Question: The carbon–carbon bond lengths in naphthalene are not equal. Use a resonance argument to explain why bond (a) is shorter than bond (b).
Question: Give the IUPAC name for each compound.
Question: Which heterocycles are aromatic?
a.
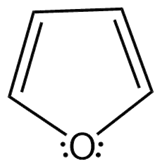
b.
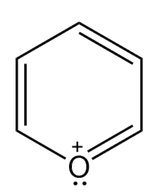
c.
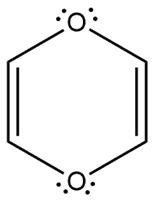
d.
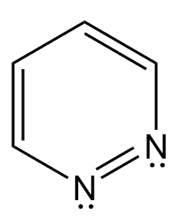
Question: Draw the four resonance structures for anthracene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.