Chapter 17: Q.46. (page 641)
Question: Which compound in each pair is the stronger acid?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 17: Q.46. (page 641)
Question: Which compound in each pair is the stronger acid?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
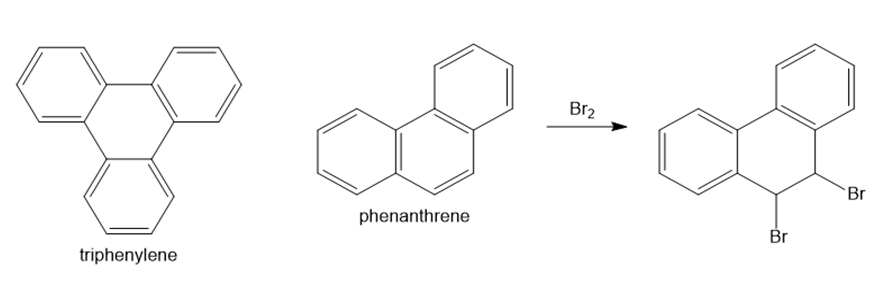
Question: Explain why triphenylene resembles benzene in that it does not undergo addition reactions with , but phenanthrene reacts with to yield the addition product drawn. (Hint: Draw resonance structures for both triphenylene and phenanthrene, and use them to determine how delocalized each bond is.)
Question: Draw the seven resonance structures for the tropyllium cation.
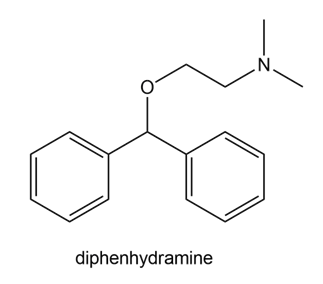
Question: Draw all possible resonance structures for the antihistamine diphenhydramine, the active ingredient in Benadryl.
Give the IUPAC name for each compound.
(a)
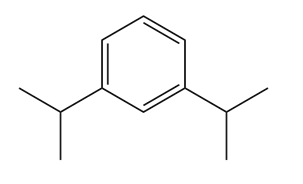
(b)
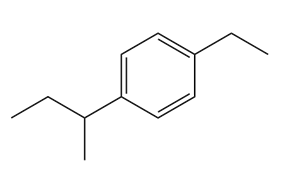
(c)
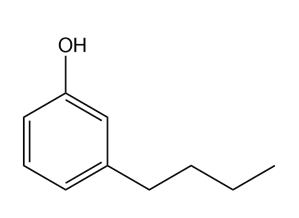
(d)
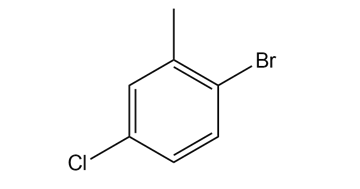
Question:
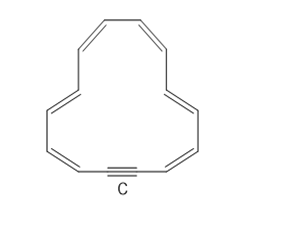
a. How many electrons does C contain?
b. How many electrons are delocalized in the ring?
c. Explain why C is aromatic.
What do you think about this solution?
We value your feedback to improve our textbook solutions.