Chapter 17: Q.1. (page 641)
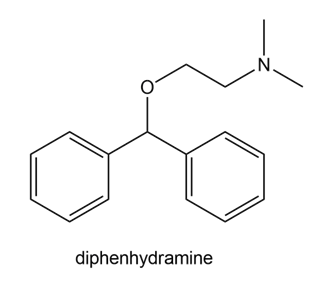
Question: Draw all possible resonance structures for the antihistamine diphenhydramine, the active ingredient in Benadryl.
Short Answer
Answer
Resonating structures of diphenhydramine.
Learning Materials
Features
Discover
Chapter 17: Q.1. (page 641)
Question: Draw all possible resonance structures for the antihistamine diphenhydramine, the active ingredient in Benadryl.

Answer
Resonating structures of diphenhydramine.

All the tools & learning materials you need for study success - in one app.
Get started for free
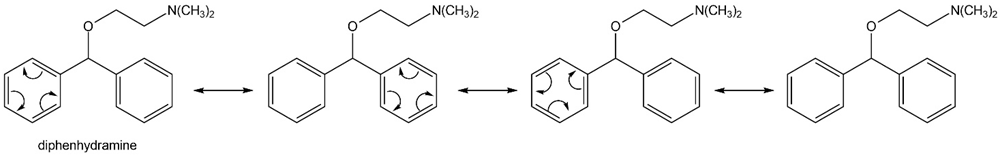
Question: Pentalene, azulene, and heptalene are conjugated hydrocarbons that do not contain a benzene ring. Which hydrocarbons are especially stable or unstable based on the number of electrons they contain? Explain your choices.
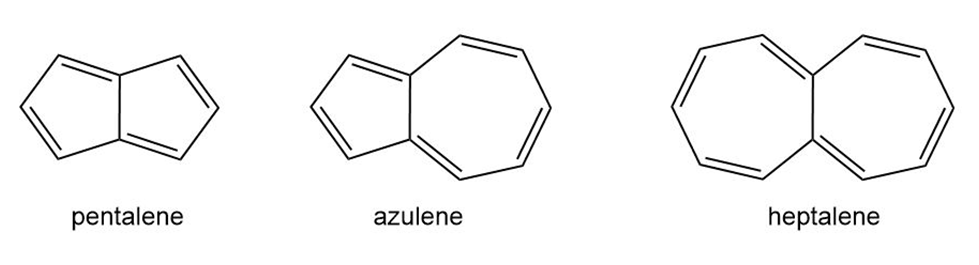
Question: Draw a stepwise mechanism for the following reaction.
Question: How many NMR signals does each compound exhibit?
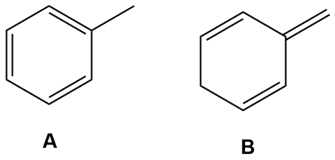
Question: You have a sample of a compound of molecular formula , which has a benzene ring substituted by two groups, and , and exhibits the given NMR. What disubstituted benzene isomer corresponds to these data?
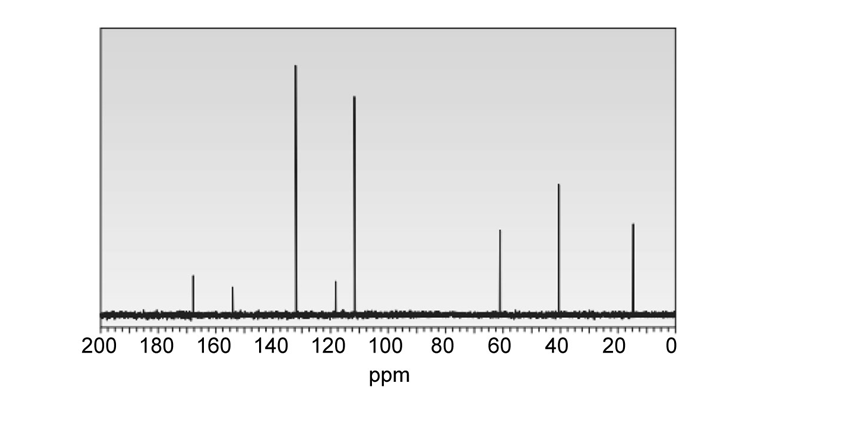
Question: Compounds A and B are both hydrogenated to methylcyclohexane. Which compound has the larger heat of hydrogenation? Which compound is more stable?
What do you think about this solution?
We value your feedback to improve our textbook solutions.