Chapter 17: Q.41. (page 641)
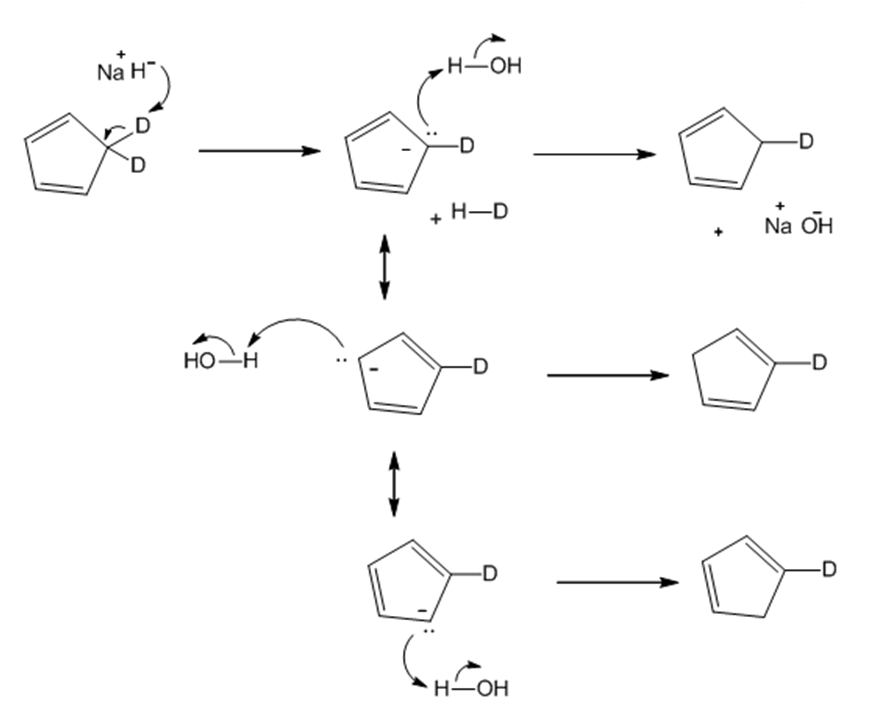
Question: Draw a stepwise mechanism for the following reaction.
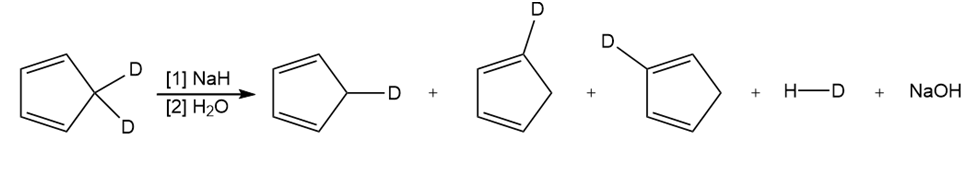
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 17: Q.41. (page 641)
Question: Draw a stepwise mechanism for the following reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use the inscribed polygon method to show the pattern of molecular orbitals in cyclooctatetraene.
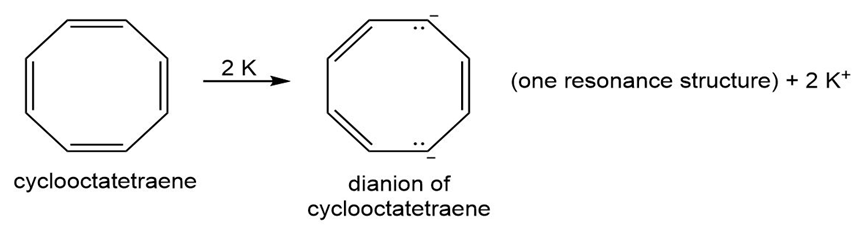
a. Label the MOs as bonding, antibonding, or nonbonding.
b. Indicate the arrangement of electrons in these orbitals for cyclooctatetraene, and explain why cyclooctatetraene is not aromatic.
c. Treatment of cyclooctatetraene with potassium forms a dianion. How manyelectrons does this dianion contain?
d. How are the electrons in this dianion arranged in the molecular orbitals?
e. Classify the dianion of cyclooctatetraene as aromatic, antiaromatic, or not aromatic, and explain why this is so.
Question: Which compound in each pair is the stronger acid?
What orbitals are used to form the labeled bonds in the following molecule? Of the labeled bonds, which is the shortest?
Question: Early structural studies on benzene had to explain the following experimental evidence. When benzene was treated with (plus a Lewis acid), a single substitution product of molecular formula was formed. When this product was treated with another equivalent of , three different compounds of molecular formularole="math" localid="1648727281591" were formed.
Question: You have a sample of a compound of molecular formula , which has a benzene ring substituted by two groups, and , and exhibits the given NMR. What disubstituted benzene isomer corresponds to these data?
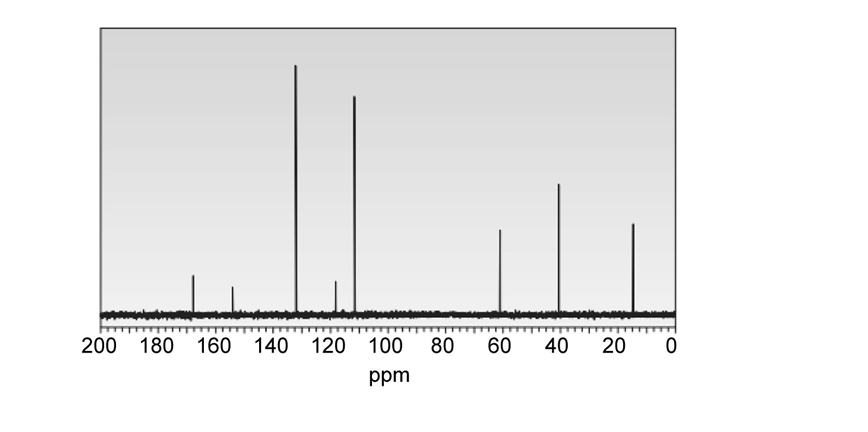
What do you think about this solution?
We value your feedback to improve our textbook solutions.