Chapter 17: Q.40. (page 641)
Question: Explain the observed rate of reactivity of the following alkyl halides in an reaction.
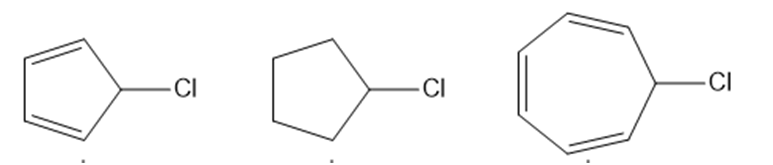
Short Answer
Answer

Learning Materials
Features
Discover
Chapter 17: Q.40. (page 641)
Question: Explain the observed rate of reactivity of the following alkyl halides in an reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Rank the following compounds in the order of increasing acidity.
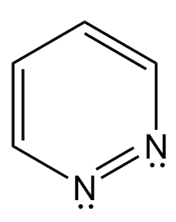
Question: Which heterocycles are aromatic?
a.
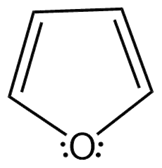
b.
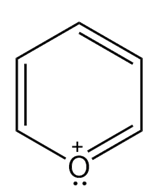
c.
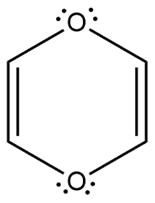
d.
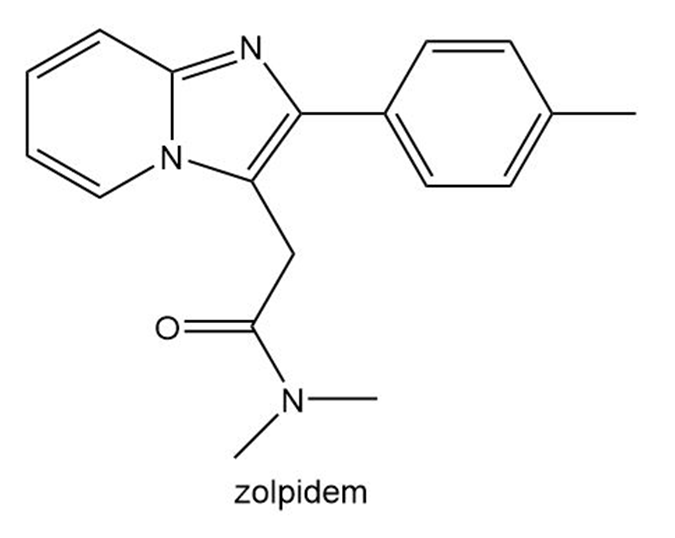
Question: Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia.
a. In what type of orbital does the lone pair on each N atom in the heterocycle reside?
b. Explain why the bicyclic ring system that contains both N atoms is aromatic.
c. Draw all reasonable resonance structures for the bicyclic ring system.
Question: Answer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.
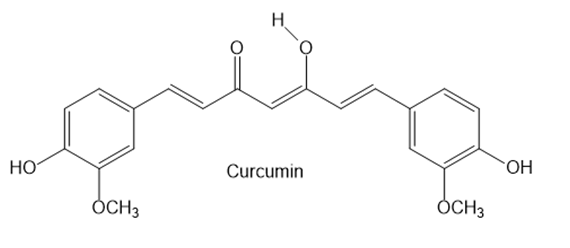
a. In Chapter 11 we learned that most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols.
b. Explain why the enol O-H proton is more acidic than an alcohol O-H proton.
c. Why is curcumin colored?
d. Explain why curcumin is an antioxidant.
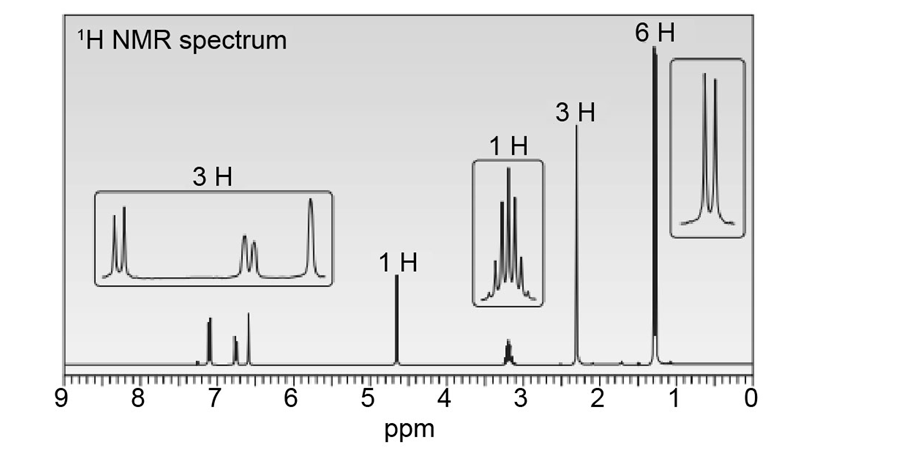
Question: Thymol (molecular formula ) is the major component of the oil of thyme. Thymol shows IR absorptions at 3500–3200, 3150–2850, 1621, and 1585 localid="1648806094089" . The NMR spectrum of thymol is given below. Propose a possible structure for thymol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.