Chapter 17: Q.16. (page 641)
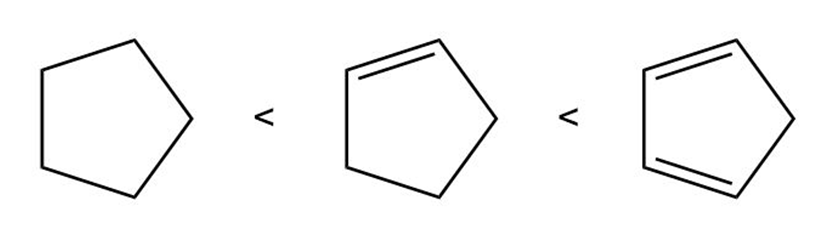
Question: Rank the following compounds in the order of increasing acidity.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 17: Q.16. (page 641)
Question: Rank the following compounds in the order of increasing acidity.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which compound in each pair is the stronger acid?
Question: What orbitals are used to form the labeled bonds in the following molecule? Of the labeled bonds, which is the shortest?
Question: Early structural studies on benzene had to explain the following experimental evidence. When benzene was treated with (plus a Lewis acid), a single substitution product of molecular formula was formed. When this product was treated with another equivalent of , three different compounds of molecular formularole="math" localid="1648727281591" were formed.
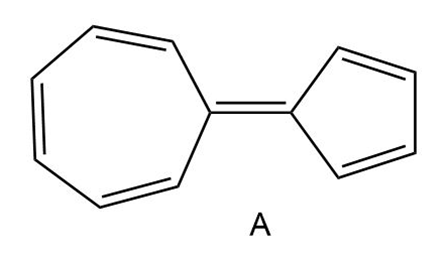
Question: Hydrocarbon A possesses a significant dipole, even though it is composed of only C-C and C-H bonds. Explain why the dipole arises and use resonance structures to illustrate the direction of the dipole. Which ring is more electron rich?
Question: Draw the structure corresponding to each name
What do you think about this solution?
We value your feedback to improve our textbook solutions.