Chapter 17: Q.34. (page 641)
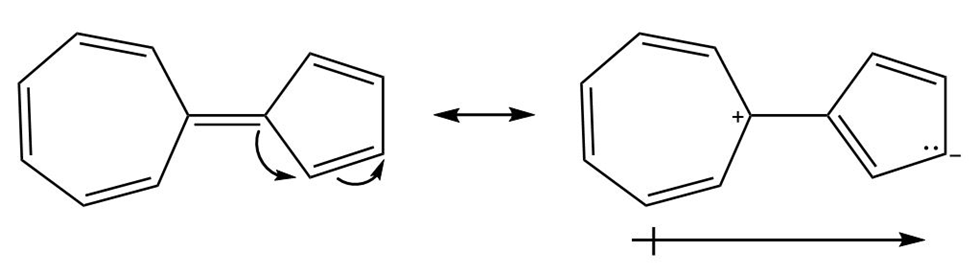
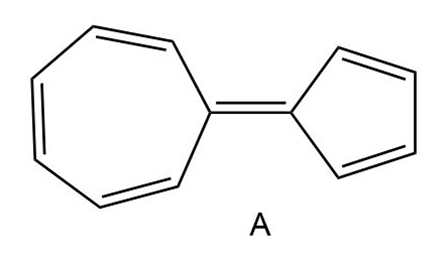
Question: Hydrocarbon A possesses a significant dipole, even though it is composed of only C-C and C-H bonds. Explain why the dipole arises and use resonance structures to illustrate the direction of the dipole. Which ring is more electron rich?
Short Answer
Answer
This molecule also possesses a net dipole moment. The seven membered ring is electron deficient and the five-membered ring is electron rich in nature as shown in the structure below:
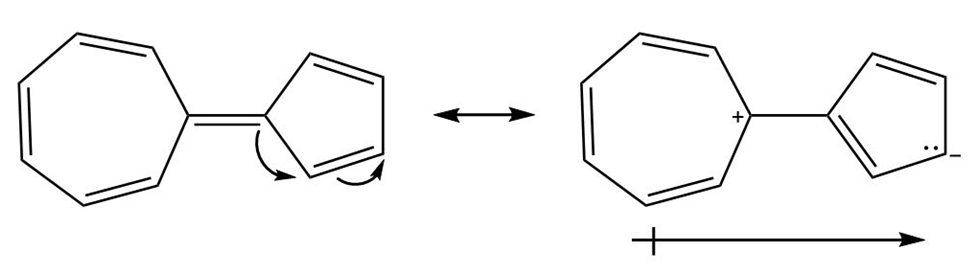
Resonating structures