Chapter 17: Q.5. (page 641)
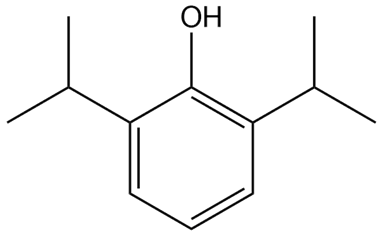
Question: What is the structure of propofol, which has the IUPAC name 2,6-diisopropylphenol? Propofol is an intravenous medication used to induce and maintain anesthesia.
Short Answer
Answer
Structure of propofol
Learning Materials
Features
Discover
Chapter 17: Q.5. (page 641)
Question: What is the structure of propofol, which has the IUPAC name 2,6-diisopropylphenol? Propofol is an intravenous medication used to induce and maintain anesthesia.
Answer
Structure of propofol

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use the inscribed polygon method to show why the cyclopentadienyl cation and radical are not aromatic.
Question: Explain why tetrahydrofuran has a higher boiling point and is much more water soluble than furan, even though both compounds are cyclic ethers containing four carbons.

Question:
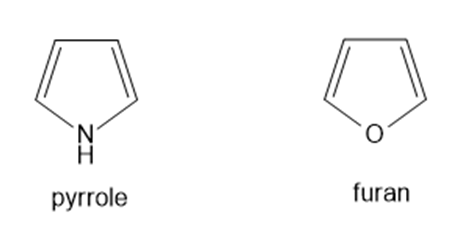
a. Draw all reasonable resonance structures for pyrrole and explain why pyrrole is less resonance stabilized than benzene.
b. Draw all reasonable resonance structures for furan and explain why furan is less resonance stabilized than pyrrole.
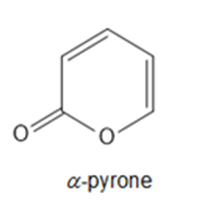
Question: Explain why -pyrone reacts with to yield a substitution product (like benzene does),rather than an addition product to one of its bonds.
Question: How many NMR signals do each compound exhibit?
a.
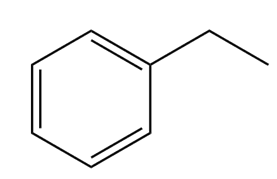
b.
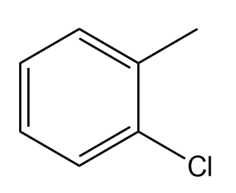
c.
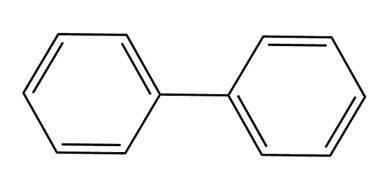
What do you think about this solution?
We value your feedback to improve our textbook solutions.