Chapter 17: Q.21. (page 641)
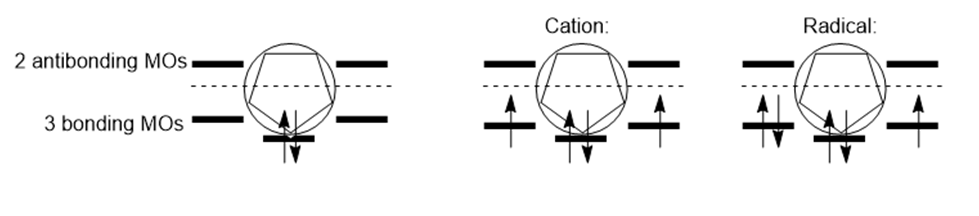
Question: Use the inscribed polygon method to show why the cyclopentadienyl cation and radical are not aromatic.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 17: Q.21. (page 641)
Question: Use the inscribed polygon method to show why the cyclopentadienyl cation and radical are not aromatic.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
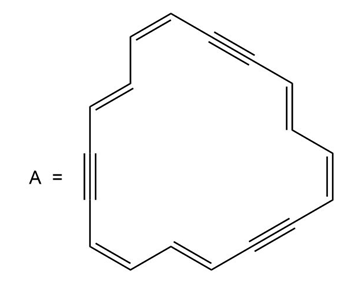
Question: Compound A exhibits a peak in its NMR spectrum at 7.6 ppm, indicating that it is aromatic.
(a) How are the carbon atoms of the triple bonds hybridized?
(b) In what type of orbitals are the electrons of the triple bonds contained?
(c) How many electrons are delocalized around the ring in A?
Question: Rank the following compounds in the order of increasing acidity.
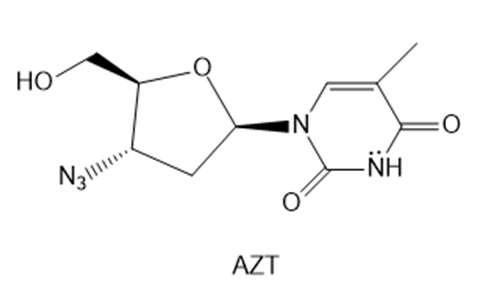
Question: AZT was the first drug approved to treat HIV, the virus that causes AIDS. Explain why the six-membered ring of AZT is aromatic.
Question: Classify each compound as aromatic, antiaromatic, or not aromatic.
Give the IUPAC name for each compound.
(a)
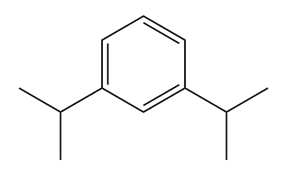
(b)
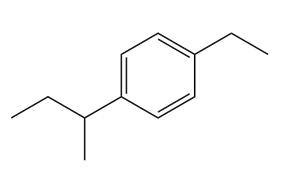
(c)
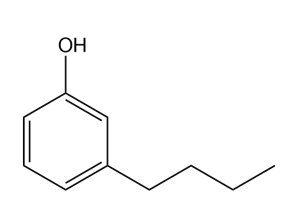
(d)
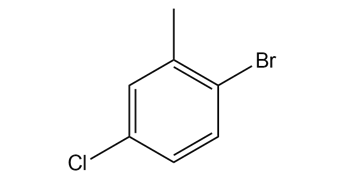
What do you think about this solution?
We value your feedback to improve our textbook solutions.