Chapter 17: Problem 17.3 (page 646)
Give the IUPAC name for each compound.
(a)
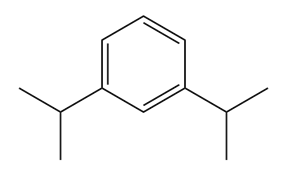
(b)
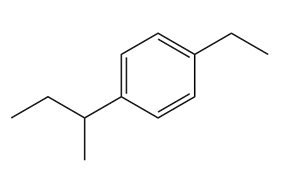
(c)
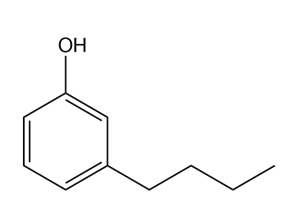
(d)
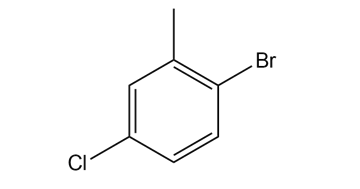
Short Answer
Answer
- m-1,3-diisopropylbenzene
- 1-sec-butyl-4-ethylbenzene
- m-butylphenol
- 2-bromo-5-chlorotoulene
Learning Materials
Features
Discover
Chapter 17: Problem 17.3 (page 646)
Give the IUPAC name for each compound.
(a)

(b)

(c)

(d)

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the diethylbenzene isomers (ortho, meta, or para) corresponds to each set of NMR spectral data?
[A] NMR signals: 16, 29, 125, 127.5, 128.4, and 144 ppm
[B] NMR signals: 15, 26, 126, 128, and 142 ppm
[C] NMR signals: 16, 29, 128, and 141 ppm
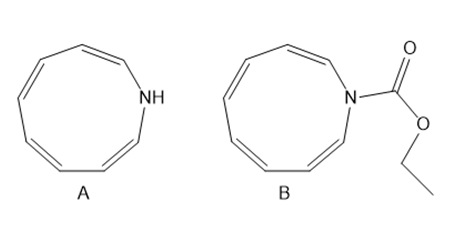
Question: Explain why A is aromatic but B is not aromatic.
Question: Use the inscribed polygon method to show why the cyclopentadienyl cation and radical are not aromatic.
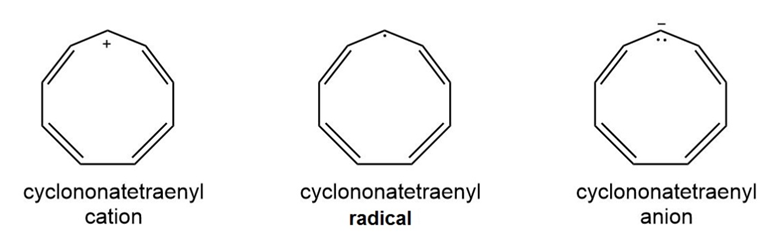
Question: Use the inscribed polygon method to show the pattern of molecular orbitals in cyclonona-1,3,5,7-tetraene and use it to label its cation, radical, and anion as aromatic, antiaromatic, or not aromatic.
Question: Draw the seven resonance structures for the tropyllium cation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.