Chapter 17: Q.12. (page 641)
Question: Which heterocycles are aromatic?
a.
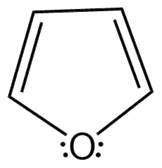
b.
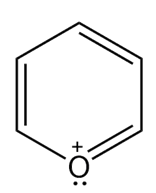
c.
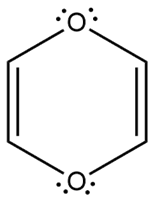
d.
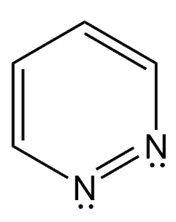
Short Answer
Answer
- Aromatic
- Aromatic
- Non-aromatic
- Aromatic
Learning Materials
Features
Discover
Chapter 17: Q.12. (page 641)
Question: Which heterocycles are aromatic?
a.

b.

c.

d.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Propose a structure consistent with each set of data.
a. : IR absorptions at ,, and
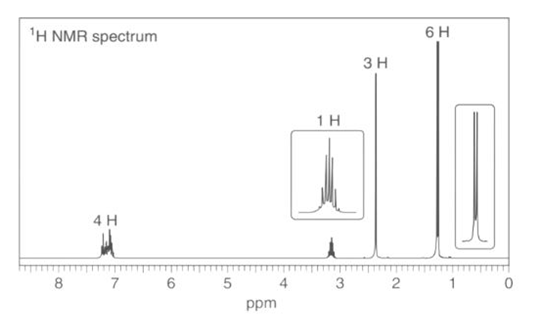
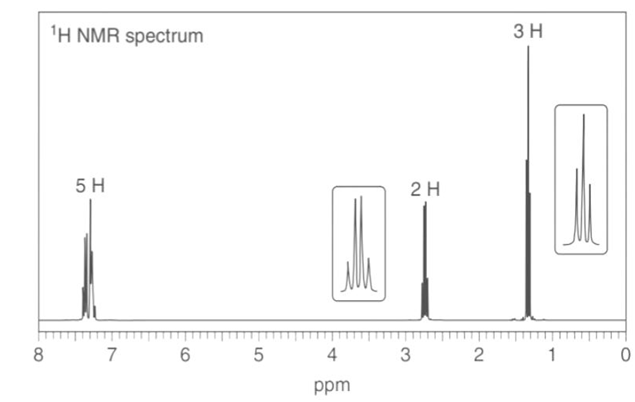
b. : NMR signals at 21, 127, and 138 ppm
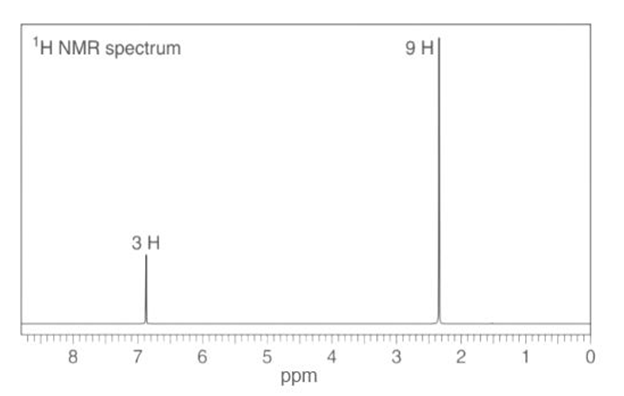
c. : IR absorptions at , 1606, and
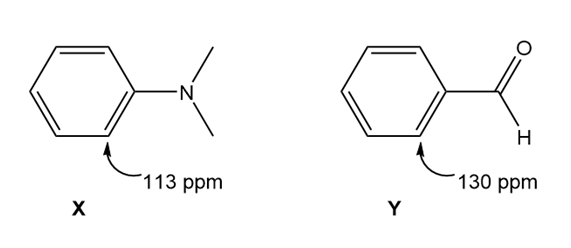
Question: Although benzene itself absorbs at 128 ppm in its NMR spectrum, the carbons of substituted benzenes absorb either upfield or downfield from this value depending on the substituent. Explain the observed values for the carbon ortho to the given substituent in the monosubstituted benzene derivatives X and Y.
Question: a. Draw the 14 constitutional isomers of molecular formula that contain a benzene ring.
b. Name all compounds that contain a trisubstituted benzene ring.
c. For which compound(s) are stereoisomers possible? Draw all possible stereoisomers.
Question: Early structural studies on benzene had to explain the following experimental evidence. When benzene was treated with (plus a Lewis acid), a single substitution product of molecular formula was formed. When this product was treated with another equivalent of , three different compounds of molecular formularole="math" localid="1648727281591" were formed.
Question: Label each compound as aromatic, antiaromatic, or not aromatic. Assume all completely conjugated rings are planar.
What do you think about this solution?
We value your feedback to improve our textbook solutions.