Chapter 17: Q.13. (page 641)
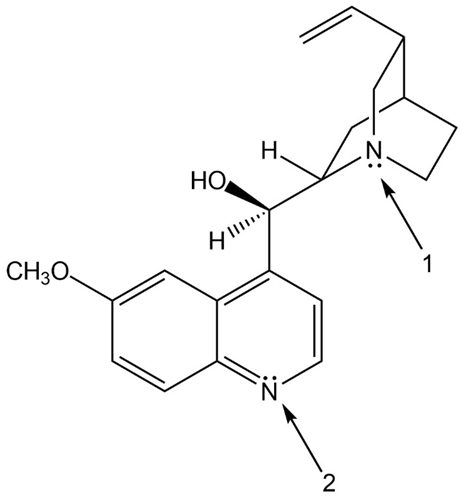
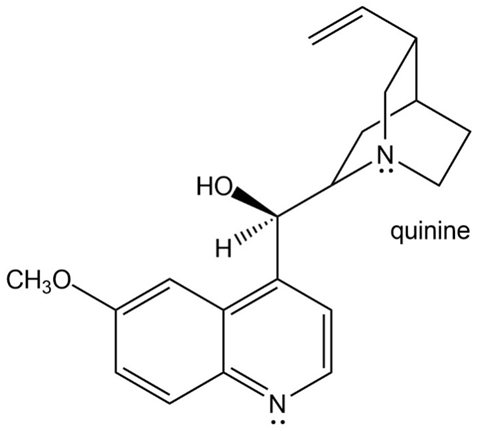
Question: (a) How is each N atom in quinine, an effective antimalarial drug that reduces fever, hybridized? (b) In what type of orbital does the lone pair on each N reside?
Short Answer
Answer
a. Hybridization of each N atom is as follows:
At 1: hybridized
At 2: hybridized
b. The lone pair resides in the following position:
At 1: hybrid orbital
At 2: hybrid orbital