Chapter 17: Q.11. (page 641)
Question: Draw the four resonance structures for anthracene.
Short Answer
Answer
Resonating structures of anthracene

Learning Materials
Features
Discover
Chapter 17: Q.11. (page 641)
Question: Draw the four resonance structures for anthracene.
Answer
Resonating structures of anthracene

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following heterocycles are aromatic?
Draw the structure corresponding to each name
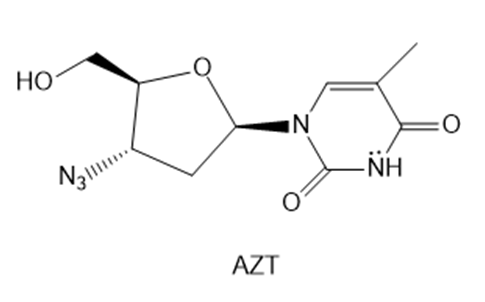
Question: AZT was the first drug approved to treat HIV, the virus that causes AIDS. Explain why the six-membered ring of AZT is aromatic.
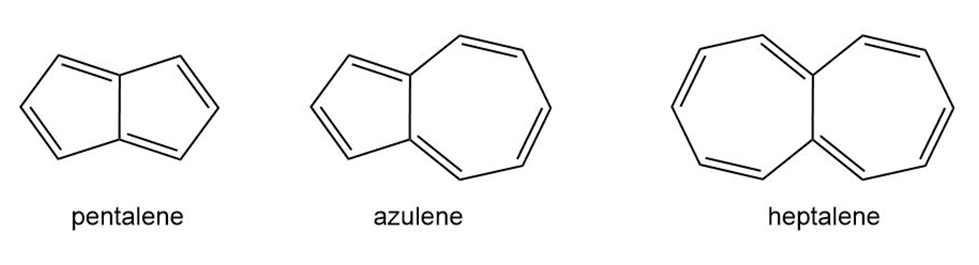
Question: Pentalene, azulene, and heptalene are conjugated hydrocarbons that do not contain a benzene ring. Which hydrocarbons are especially stable or unstable based on the number of electrons they contain? Explain your choices.
Question: Stanozolol is an anabolic steroid that promotes muscle growth. Although stanozolol has been used by athletes and bodybuilders, many physical and psychological problems result from prolonged use and it is banned in competitive sports.
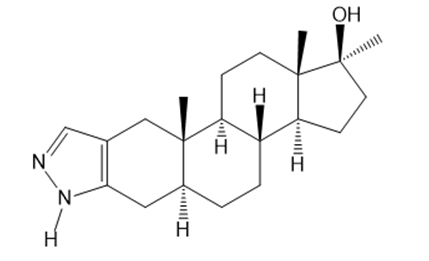
a. Explain why the nitrogen heterocycle—a pyrazole ring—is aromatic.
b. In what type of orbital is the lone pair on each N atom contained?
c. Draw all reasonable resonance structures for stanozolol.
d. Explain why the of the N-H bond in the pyrazole ring is comparable to the of O-H bond, making it considerably more acidic than amines such as ( = 40).
What do you think about this solution?
We value your feedback to improve our textbook solutions.