Chapter 17: Q.55. (page 641)
Question: Propose a structure consistent with each set of data.
a. : IR absorptions at ,, and
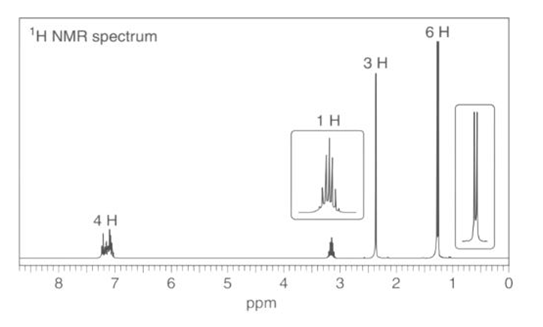
b. : NMR signals at 21, 127, and 138 ppm
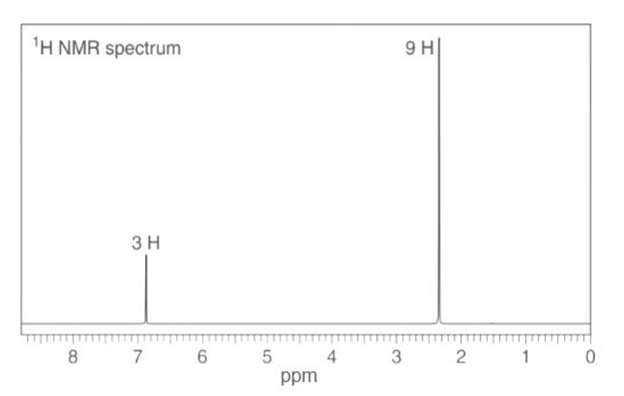
c. : IR absorptions at , 1606, and
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 17: Q.55. (page 641)
Question: Propose a structure consistent with each set of data.
a. : IR absorptions at ,, and

b. : NMR signals at 21, 127, and 138 ppm

c. : IR absorptions at , 1606, and

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Januvia, the trade name for sitagliptin, was introduced in 2006 for the treatment of type 2 diabetes. (a) Explain why the five-membered ring in sitagliptin is aromatic. (b) Determine the hybridization of each N atom. (c) In what type of orbital does the lone pair on each N atom reside?
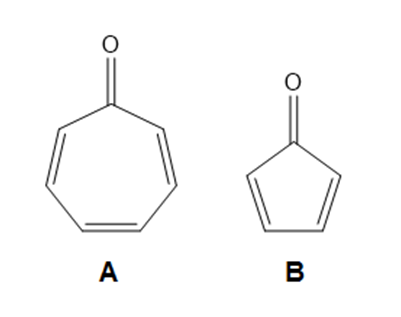
Question: Explain why compound A is much more stable than compound B.
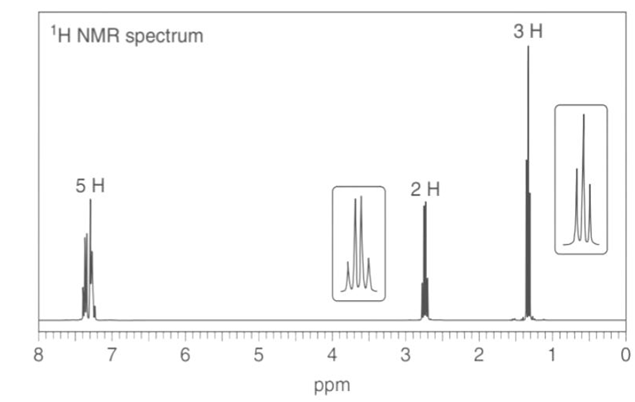
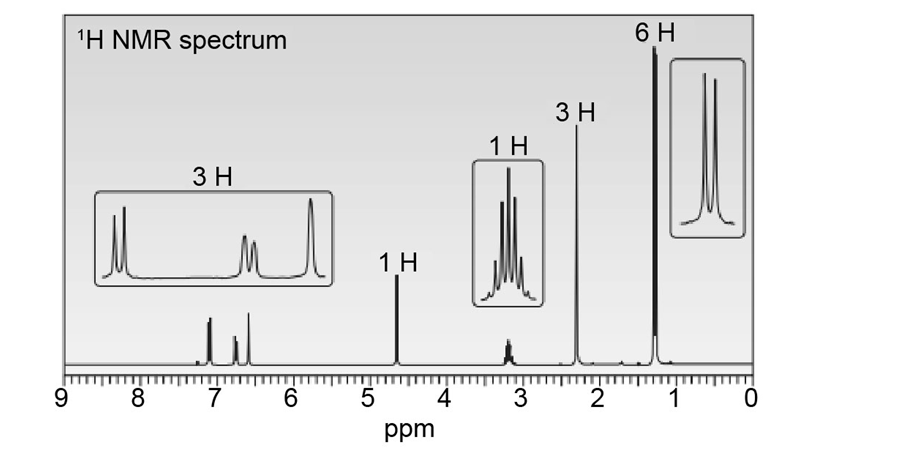
Question: Thymol (molecular formula ) is the major component of the oil of thyme. Thymol shows IR absorptions at 3500–3200, 3150–2850, 1621, and 1585 localid="1648806094089" . The NMR spectrum of thymol is given below. Propose a possible structure for thymol.
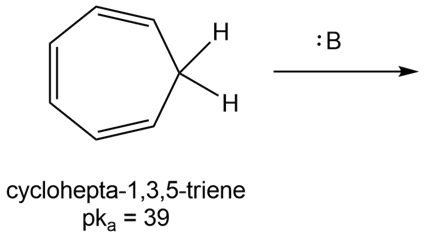
Question: Draw the product formed when cyclohepta-1,3,5-triene (pKa = 39) is treated with a strong base. Why is its pKa so much higher than the pKa of cyclopentadiene?
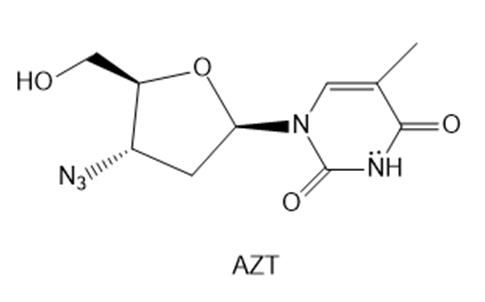
Question: AZT was the first drug approved to treat HIV, the virus that causes AIDS. Explain why the six-membered ring of AZT is aromatic.
What do you think about this solution?
We value your feedback to improve our textbook solutions.