Chapter 17: Q.66. (page 641)
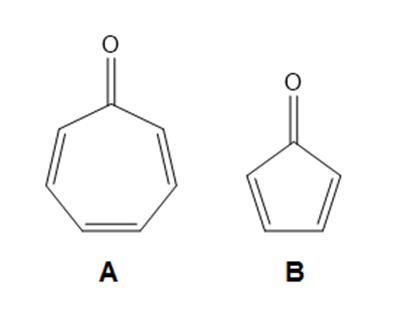
Question: Explain why compound A is much more stable than compound B.
Short Answer
Answer
The aromaticity of compound A makes it more stable than compound B.
Learning Materials
Features
Discover
Chapter 17: Q.66. (page 641)
Question: Explain why compound A is much more stable than compound B.

Answer
The aromaticity of compound A makes it more stable than compound B.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What is the structure of propofol, which has the IUPAC name 2,6-diisopropylphenol? Propofol is an intravenous medication used to induce and maintain anesthesia.
Question: Compound A exhibits a peak in its NMR spectrum at 7.6 ppm, indicating that it is aromatic.
(a) How are the carbon atoms of the triple bonds hybridized?
(b) In what type of orbitals are the electrons of the triple bonds contained?
(c) How many electrons are delocalized around the ring in A?
Question: Draw additional resonance structures for each species.
Question: How many NMR signals does each compound exhibit?
Question: What is the structure of a compound of molecular formula that shows a strong IR absorption at 3150–2850 role="math" localid="1648710122642" and gives the following 1 H NMR absorptions: 1.4 (triplet, 6 H), 4.0 (quartet, 4 H), and 6.8 (singlet, 4 H) ppm
What do you think about this solution?
We value your feedback to improve our textbook solutions.