Chapter 17: Q.43. (page 641)
Question: Draw additional resonance structures for each species.
Short Answer
Answer
a.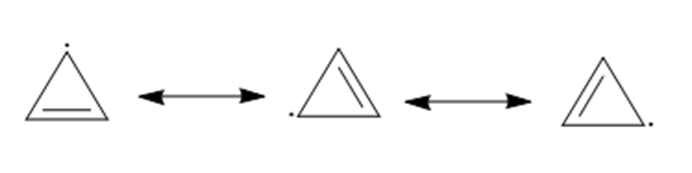
b.
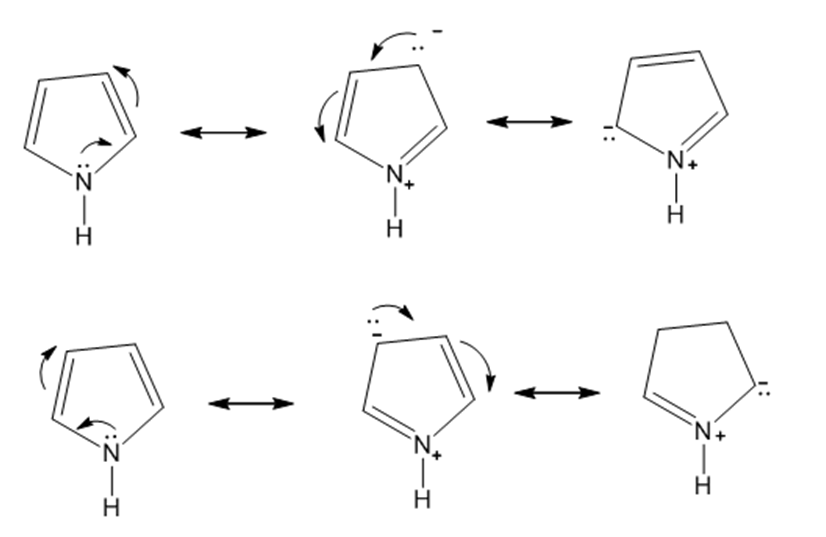
c.
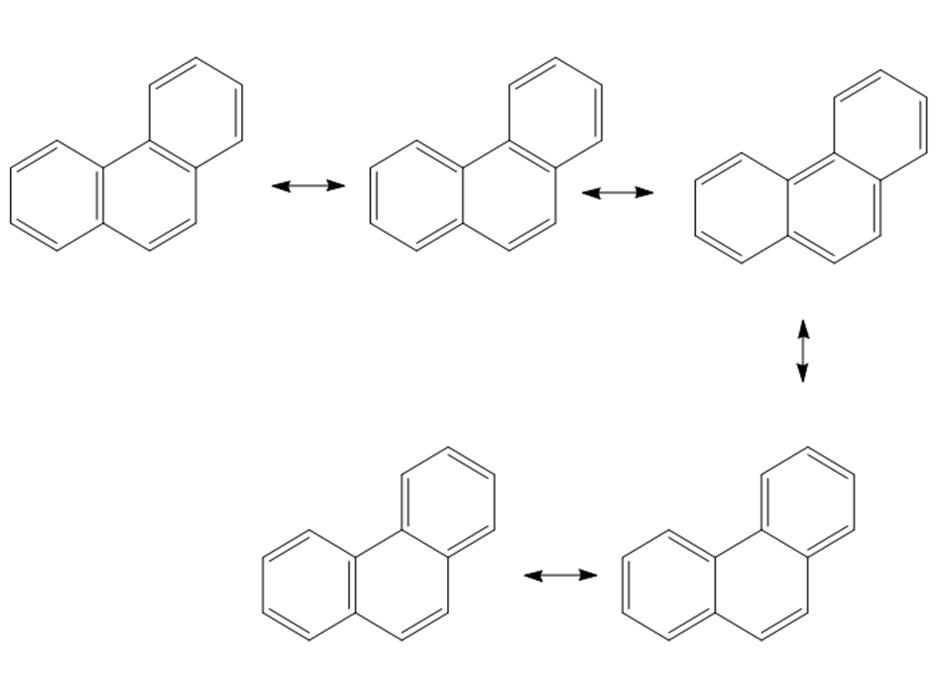
Learning Materials
Features
Discover
Chapter 17: Q.43. (page 641)
Question: Draw additional resonance structures for each species.
Answer
a.
b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
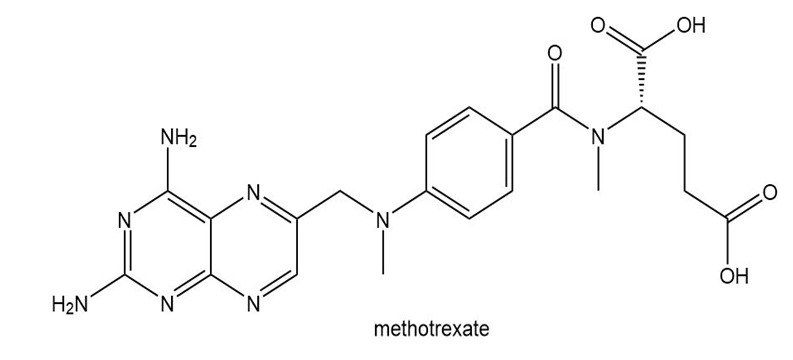
Question: Methotrexate, a drug that inhibits the metabolism of folic acid, is used in the treatment of a variety of cancers and autoimmune disorders such as rheumatoid arthritis. (a) Give the hybridization of each N atom in methotrexate. (b) In what type of orbital does the lone pair of each N reside? (c) Explain why the bicyclic ring system that contains four N atoms is aromatic.
Question: Considering both 5-methylcyclopenta-1,3-diene (A) and 7-methylcyclohepta-1,3,5-triene (B), which labeled H atom is most acidic? Which labeled H atom is least acidic? Explain your choices.
Question: Early structural studies on benzene had to explain the following experimental evidence. When benzene was treated with (plus a Lewis acid), a single substitution product of molecular formula was formed. When this product was treated with another equivalent of , three different compounds of molecular formularole="math" localid="1648727281591" were formed.
Question: Thymol (molecular formula ) is the major component of the oil of thyme. Thymol shows IR absorptions at 3500–3200, 3150–2850, 1621, and 1585 localid="1648806094089" . The NMR spectrum of thymol is given below. Propose a possible structure for thymol.
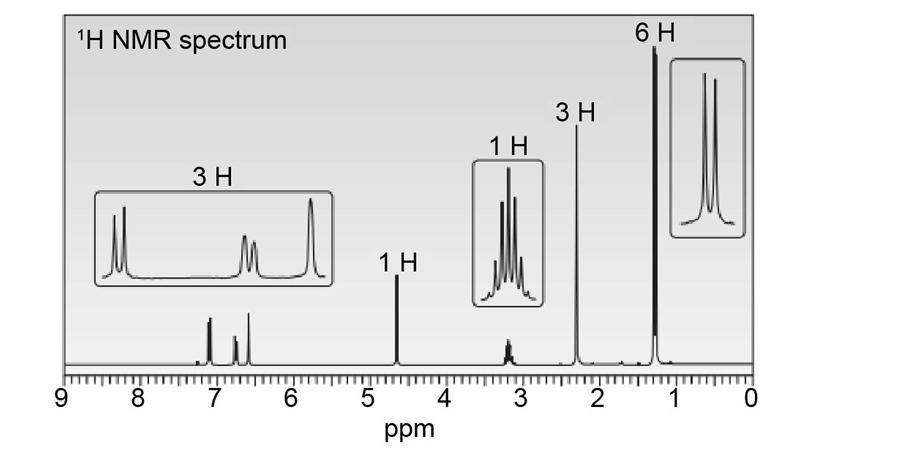
Question: Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia.
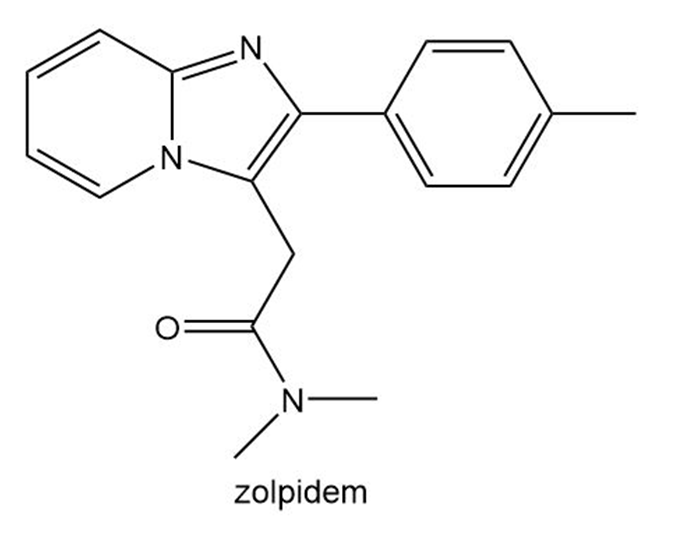
a. In what type of orbital does the lone pair on each N atom in the heterocycle reside?
b. Explain why the bicyclic ring system that contains both N atoms is aromatic.
c. Draw all reasonable resonance structures for the bicyclic ring system.
What do you think about this solution?
We value your feedback to improve our textbook solutions.