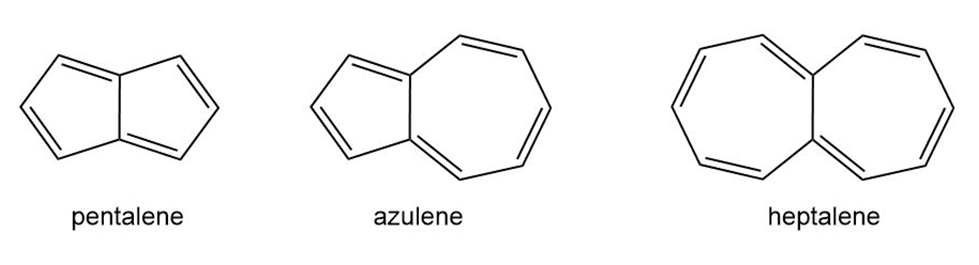
Chapter 17: Q.38. (page 641)
Question:
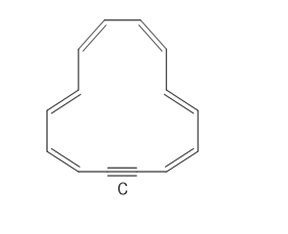
a. How many electrons does C contain?
b. How many electrons are delocalized in the ring?
c. Explain why C is aromatic.
Short Answer
Answer
a. The compound C has electrons.
b. The number of electrons delocalized in the ring is .
c. Compound C is cyclic, planar, completely conjugated and satisfies the electrons, and is aromatic.