Chapter 29: Question 29.13 (page 1164)
What alkene is needed to synthesize each amino acid by an enantioselective hydrogenation reaction using and :
(a) Alanine;
(b) Leucine;
(c) Glutamine?
Short Answer
Answers
(a)

(b)

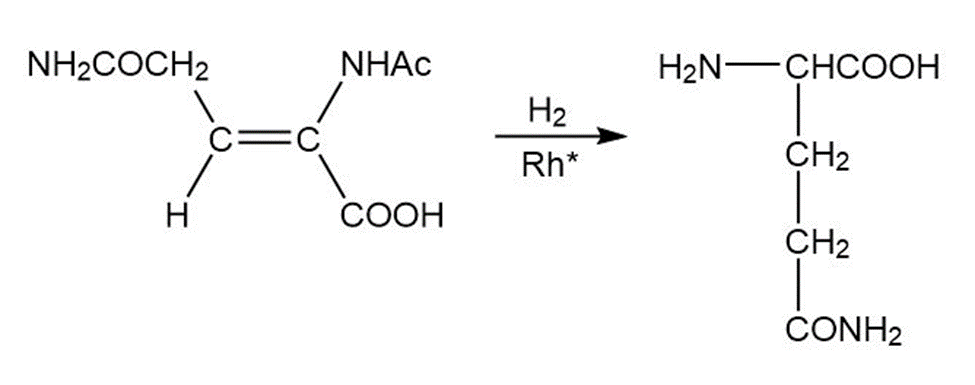
(c)
Learning Materials
Features
Discover
Chapter 29: Question 29.13 (page 1164)
What alkene is needed to synthesize each amino acid by an enantioselective hydrogenation reaction using and :
(a) Alanine;
(b) Leucine;
(c) Glutamine?
Answers
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Write out the steps for the synthesis of each peptide using the Merrifield method:
(a)Ala-Leu-Phe-Phe;
(b) Phe-Gly-Ala-Ile.
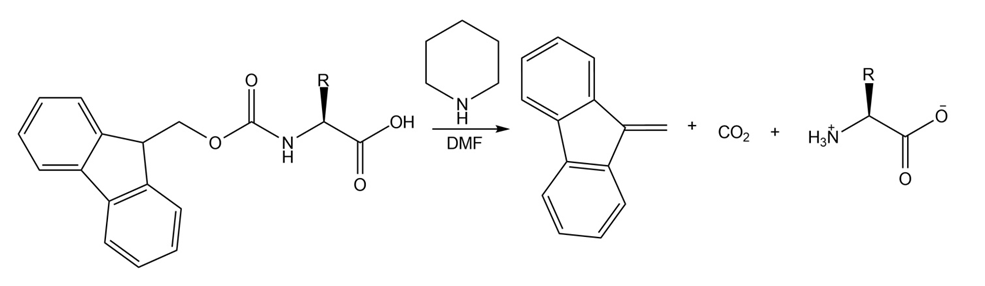
Draw the mechanism for the reaction that removes an Fmoc group from an amino acid under the following conditions:
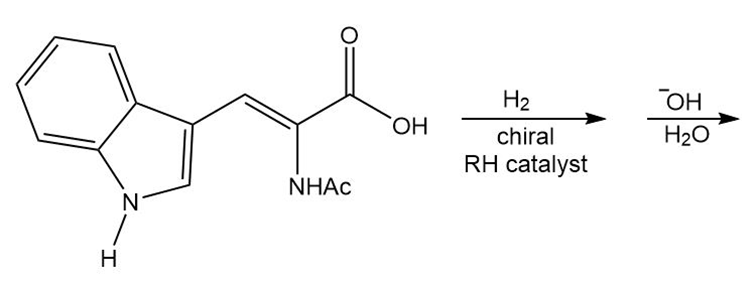
Draw the organic products formed in each reaction.
a.

b.
What form exists at the isoelectric point of each of the following amino acids: (a) valine; (b)leucine; (c)proline; (d) glutamic acid.
Draw all the steps in the synthesis of each peptide from individual amino acids:
(a)Gly-Ala;
(b) Ile-Ala-Phe
What do you think about this solution?
We value your feedback to improve our textbook solutions.