Chapter 29: 62P (page 1196)
Write out the steps for the synthesis of each peptide using the Merrifield method:
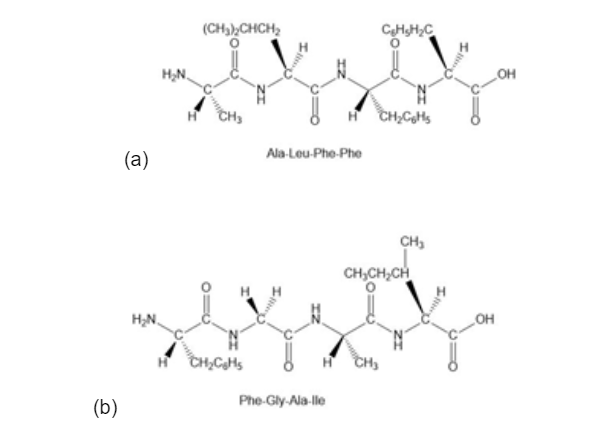
(a)Ala-Leu-Phe-Phe;
(b) Phe-Gly-Ala-Ile.
Short Answer
Learning Materials
Features
Discover
Chapter 29: 62P (page 1196)
Write out the steps for the synthesis of each peptide using the Merrifield method:
(a)Ala-Leu-Phe-Phe;
(b) Phe-Gly-Ala-Ile.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure for each peptide:
(a) Phe–Ala;
(b) Gly–Gln;
(c) Lys–Gly;
(d) R-H
Devise a synthesis of threonine from diethyl acetamidomalonate.
Consider two molecules of a tetrapeptide composed of only alanine residues. Draw the hydrogen bonding interactions that result when these two peptides adopt a parallel β-pleated sheet arrangement. Answer this same question for the antiparallel β-pleated sheet arrangement.
Besides the Boc and Fmoc protecting groups used in peptide synthesis, amines can also be protected by reaction with benzyl chloroformate (). Draw the structure of the product formed by reaction of alanine with benzyl chloroformate
Besides asymmetric hydrogenation (Section 29.4), several other methods are now available for the synthesis of optically active amino acids. How might a reaction like the Strecker synthesis be adapted to the preparation of chiral amino acids?
What do you think about this solution?
We value your feedback to improve our textbook solutions.