Chapter 29: 61P (page 1196)
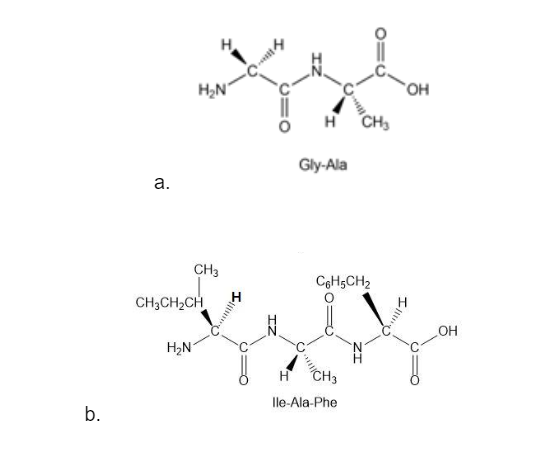
Draw all the steps in the synthesis of each peptide from individual amino acids:
Gly-Ala;
- Ile-Ala-Phe
Short Answer
Learning Materials
Features
Discover
Chapter 29: 61P (page 1196)
Draw all the steps in the synthesis of each peptide from individual amino acids:
Gly-Ala;

All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the decapeptide angiotensin I.

The larger polypeptide is angiotensin II, a hormone that narrows blood vessels and increases blood pressure. Give the amino acid sequence of angiotensin II using threeletter abbreviations. ACE inhibitors are drugs that lower blood pressure by inhibiting the ACE enzyme (Problem 5.15).
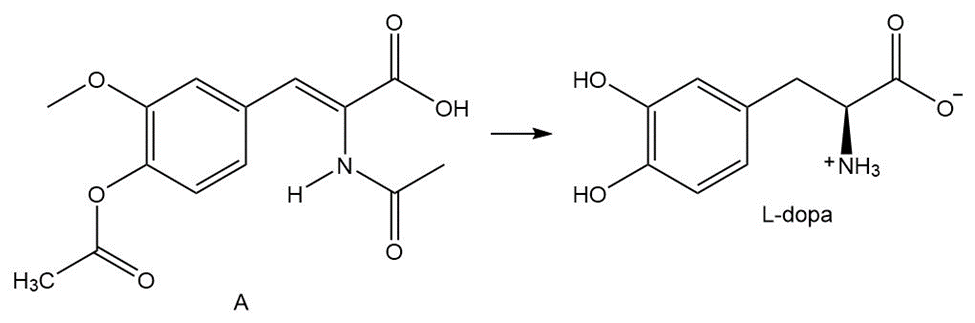
What steps are needed to convert A to L-dopa, an uncommon amino acid that is effective in treating Parkinson’s disease? These steps are the key reactions in the first commercial asymmetric synthesis using a chiral transition metal catalyst. This process was developed at Monsanto in 1974.
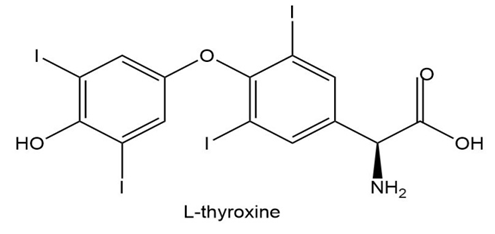
L-thyroxine, a thyroid hormone and oral medication used to treat thyroid hormone deficiency, is an amino acid that does not exist in proteins. Draw the zwitterionic form of L-thyroxine.
a.(S)-Penicillamine, an amino acid that does not occur in proteins, is used as a copper chelating agent to treat Wilson’s disease, an inherited defect in copper metabolism.
(R)-Penicillamine is toxic, sometimes causing blindness. Draw the structures of (R) and (S)-penicillamine.
b. What disulfide is formed from oxidation of L-penicillamine?
Draw the structure for each peptide:
(a) Phe–Ala;
(b) Gly–Gln;
(c) Lys–Gly;
(d) R-H
What do you think about this solution?
We value your feedback to improve our textbook solutions.