Chapter 29: Question 29.11 (page 1162)
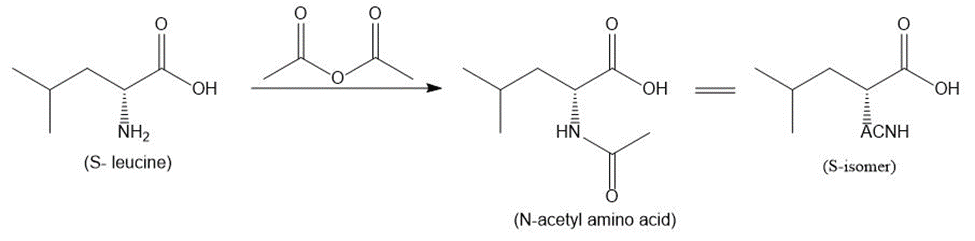
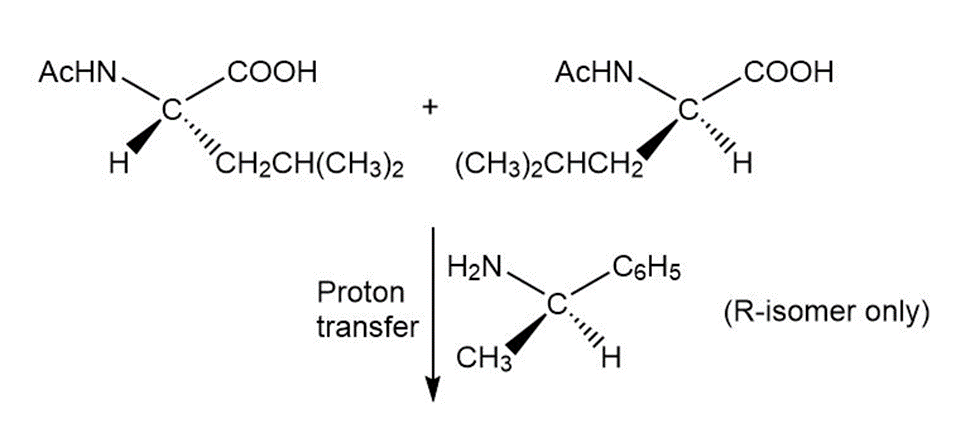
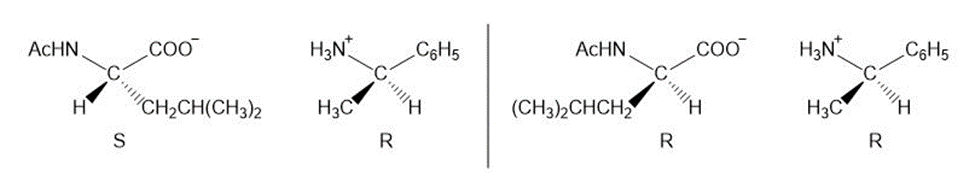
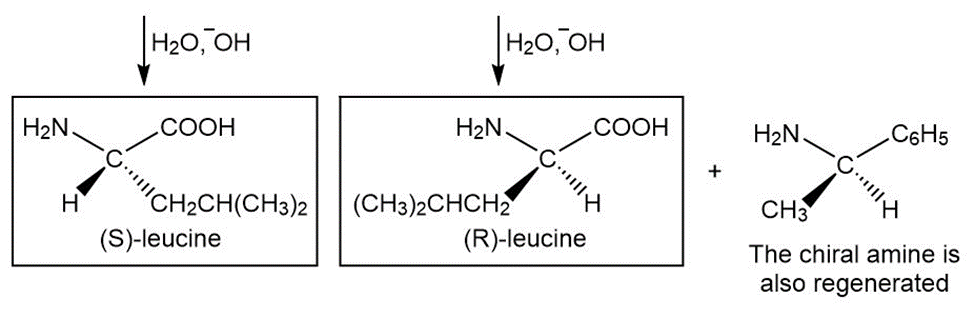
Write out a stepwise sequence that shows how a racemic mixture of leucine enantiomers can be resolved into optically active amino acids using .
Short Answer
Answer

Learning Materials
Features
Discover
Chapter 29: Question 29.11 (page 1162)
Write out a stepwise sequence that shows how a racemic mixture of leucine enantiomers can be resolved into optically active amino acids using .
Answer





All the tools & learning materials you need for study success - in one app.
Get started for free
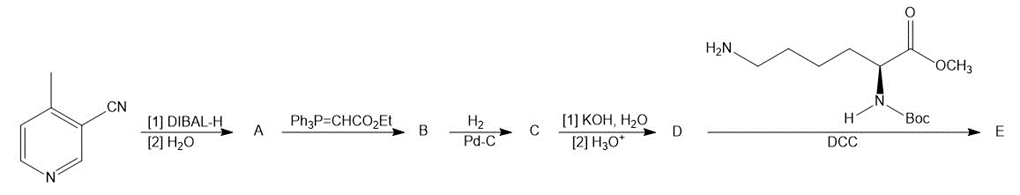
Identify A–E in the following reaction sequence.
Draw the structure of leu-enkephalin, a pentapeptide that acts as an analgesic and opiate, and has the following sequence: Tyr–Gly–Gly–Phe–Leu. (The structure of a related peptide, met-enkephalin,
appeared in Section 22.6B.)
How many different tripeptides can be formed from three different amino acids?
Which of the following amino acids are typically found in the interior of a globular protein, and which are typically found on the surface: (a)phenylalanine; (b)aspartic acid; (c) lysine; (d) isoleucine; (e) arginine; (f) glutamic acid?
Draw the organic products formed in each reaction.
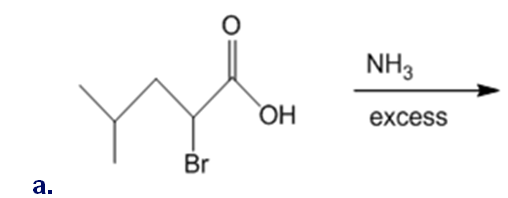
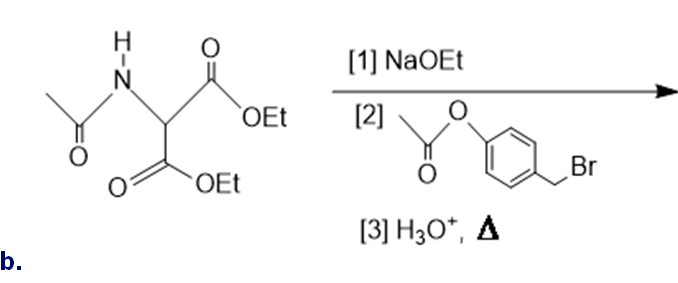
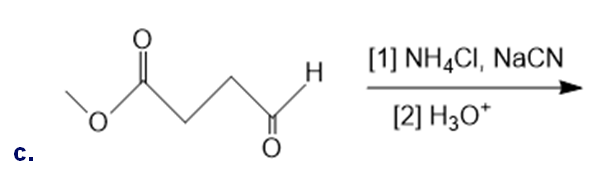
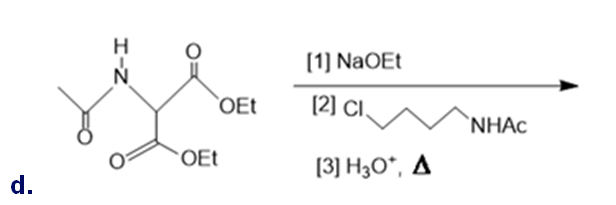
What do you think about this solution?
We value your feedback to improve our textbook solutions.