Chapter 29: Q61. (page 1196)
Draw all the steps in the synthesis of each peptide from individual amino acids:
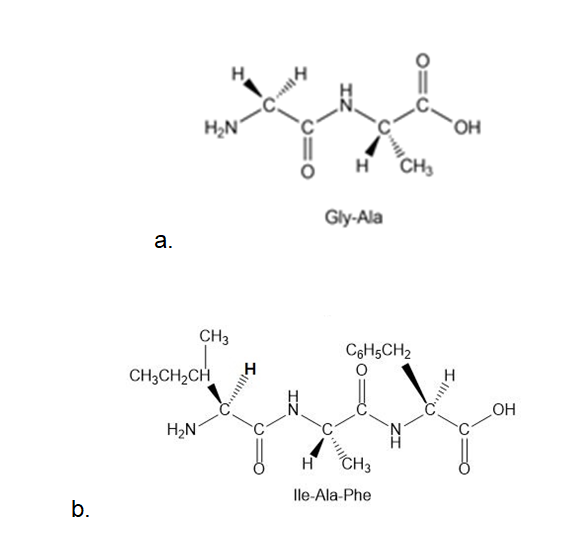
(a)Gly-Ala;
(b) Ile-Ala-Phe
Short Answer
Learning Materials
Features
Discover
Chapter 29: Q61. (page 1196)
Draw all the steps in the synthesis of each peptide from individual amino acids:
(a)Gly-Ala;
(b) Ile-Ala-Phe

All the tools & learning materials you need for study success - in one app.
Get started for free
Write out the steps for the synthesis of each peptide using the Merrifield method:
(a)Ala-Leu-Phe-Phe;
(b) Phe-Gly-Ala-Ile.
As shown in Mechanism 29.2, the final steps in the Edman degradation result in rearrangement of a thiazolinone to an N-phenylthiohydantoin. Draw a stepwise mechanism for this acid-catalyzed reaction.

Draw the mechanism for the reaction that removes an Fmoc group from an amino acid under the following conditions:

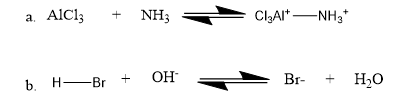
Question: Identify the nucleophile and the electrophile in the following acid–base reactions:
Write out a stepwise sequence that shows how a racemic mixture of leucine enantiomers can be resolved into optically active amino acids using .
What do you think about this solution?
We value your feedback to improve our textbook solutions.