Chapter 29: Q49P (page 1194)
Draw the organic products formed in each reaction.
a.
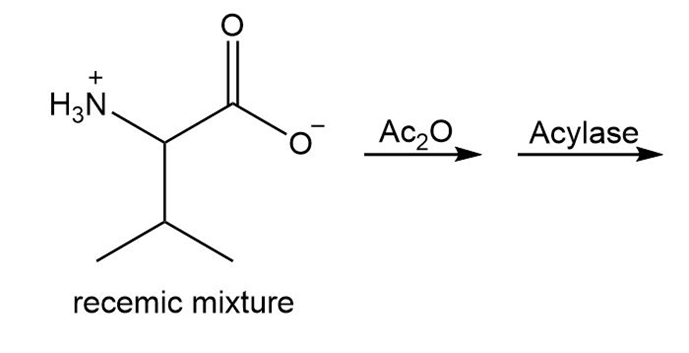
b.

Short Answer
a.
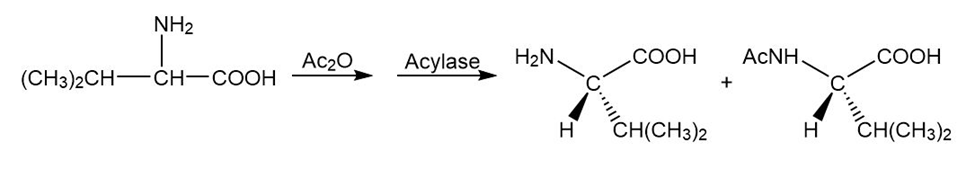
b.
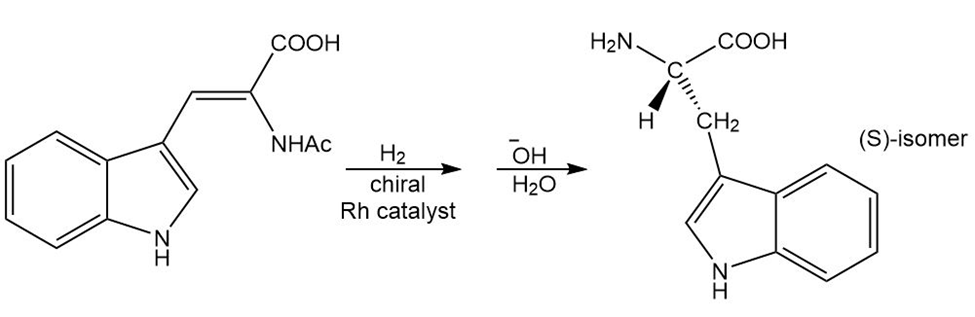
Learning Materials
Features
Discover
Chapter 29: Q49P (page 1194)
Draw the organic products formed in each reaction.
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following amino acids are typically found in the interior of a globular protein, and which are typically found on the surface: (a)phenylalanine; (b)aspartic acid; (c) lysine; (d) isoleucine; (e) arginine; (f) glutamic acid?
a. Draw the structure of the tripeptide A-A-A, and label the two ionizable functional groups.
b. What is the predominant form of A-A-A at pH=1?
c. The values for the two ionizable functional groups (3.39 and 8.03) differ considerably from the values of alanine (2.35 and 9.87;see table 29.1). Account for the observed differences.
What steps are needed to convert A to L-dopa, an uncommon amino acid that is effective in treating Parkinson’s disease? These steps are the key reactions in the first commercial asymmetric synthesis using a chiral transition metal catalyst. This process was developed at Monsanto in 1974.
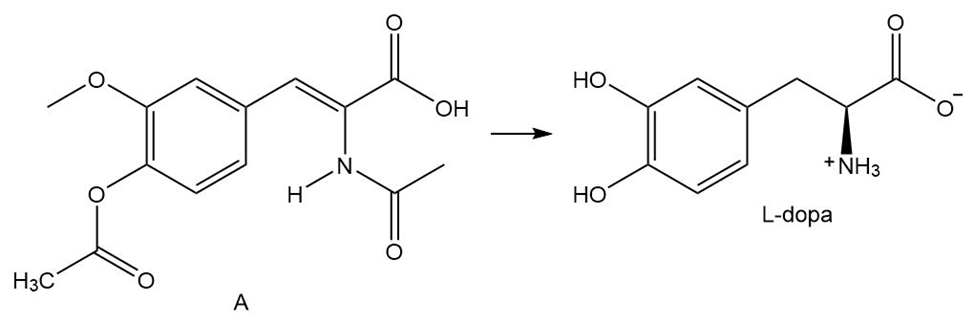
Outline the steps needed to synthesize the tetrapeptideAla–Leu–Ile–Gly using the Merrifield technique.
How many different tripeptides can be formed from three different amino acids?
What do you think about this solution?
We value your feedback to improve our textbook solutions.