Chapter 29: Q8. (page 1158)
What aldehyde is needed to synthesize each amino acid by the Strecker synthesis?
(a) Valine;
(b) Leucine;
(c) Phenylalanine.
Short Answer
(a)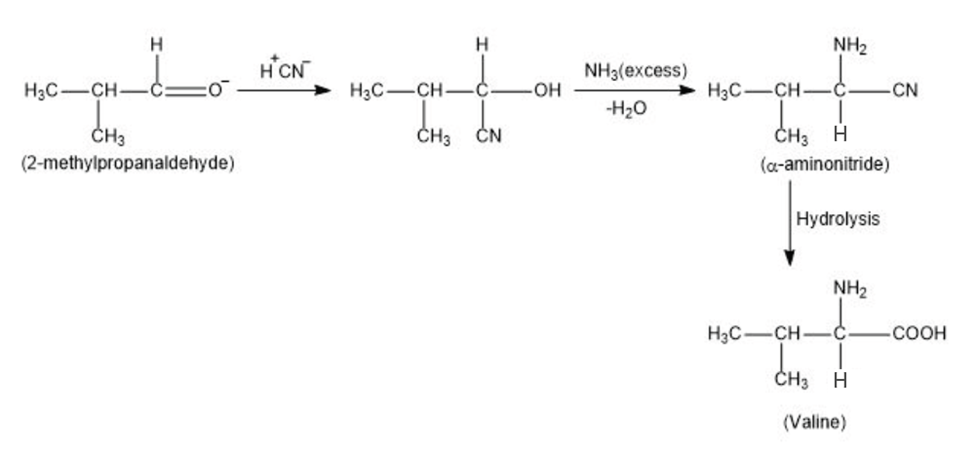
(b)
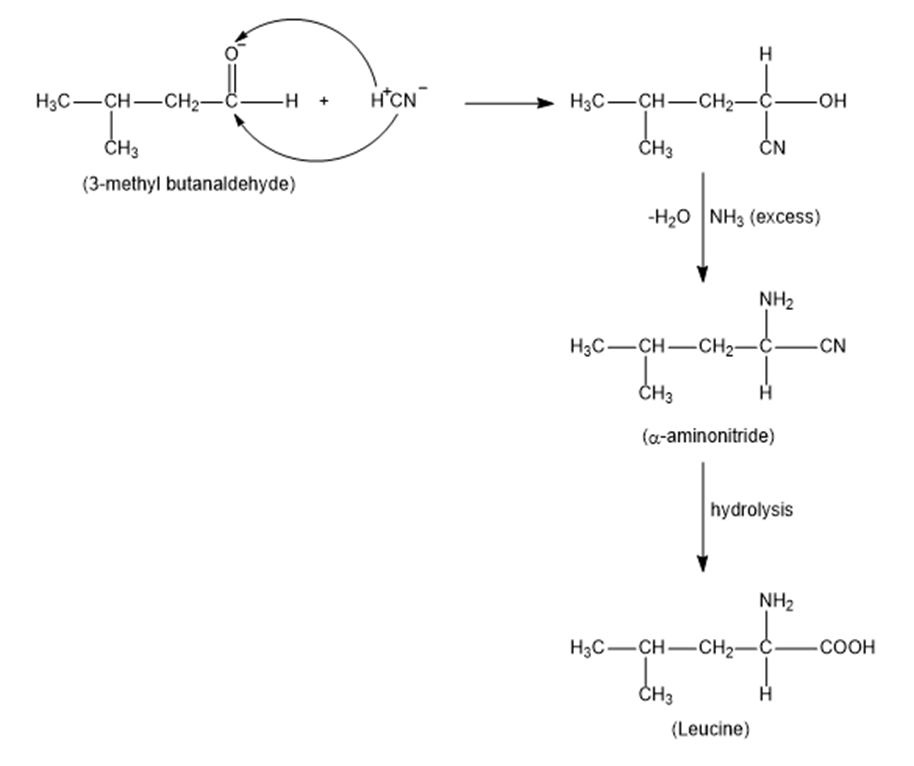
(c)
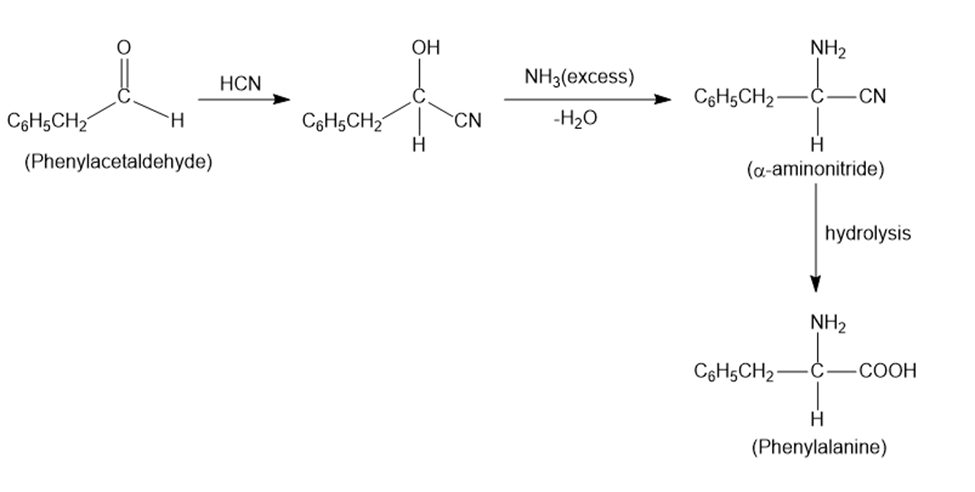
Learning Materials
Features
Discover
Chapter 29: Q8. (page 1158)
What aldehyde is needed to synthesize each amino acid by the Strecker synthesis?
(a) Valine;
(b) Leucine;
(c) Phenylalanine.
(a)
(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the organic products formed in each reaction.
a.
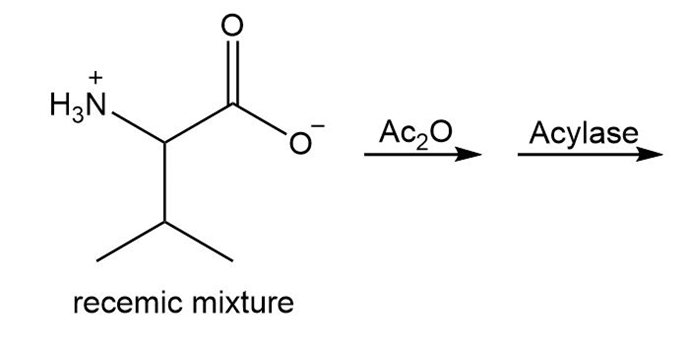
b.

The enolate derived from diethyl acetamidomalonate is treated with each of the following alkyl halides. After hydrolysis and decarboxylation, what amino acid is formed?
Histidine is classified as a basic amino acid because one of the N atoms in its five-membered ring is readily protonated by acid. Which N atom in histidine is protonated and why?
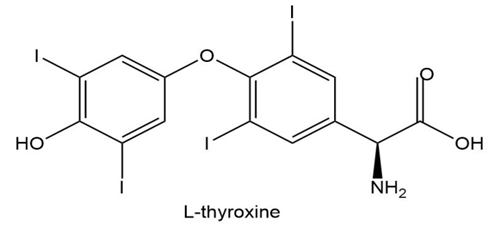
L-thyroxine, a thyroid hormone and oral medication used to treat thyroid hormone deficiency, is an amino acid that does not exist in proteins. Draw the zwitterionic form of L-thyroxine.
Propose a structure consistent with each set of data.
a. Compound J: molecular ion at 72; IR peak at 1710 \({\bf{c}}{{\bf{m}}^{{\bf{ - 1}}}}\); \({}^{\bf{1}}{\bf{H}}\)-NMR data (ppm) at 1.0 (triplet, 3 H), 2.1 (singlet, 3 H), and 2.4 (quartet, 2 H)
b. Compound K: molecular ion at 88; IR peak at 3600–3200 \({\bf{c}}{{\bf{m}}^{{\bf{ - 1}}}}\); \({}^{\bf{1}}{\bf{H}}\)-NMR data (ppm) at 0.9 (triplet, 3 H), 1.2 (singlet, 6 H), 1.5 (quartet, 2 H), and 1.6 (singlet, 1 H)
What do you think about this solution?
We value your feedback to improve our textbook solutions.