Chapter 4: Q.50. (page 128)
Question: Calculate the barrier to rotation for each bond highlighted in pink.
a.
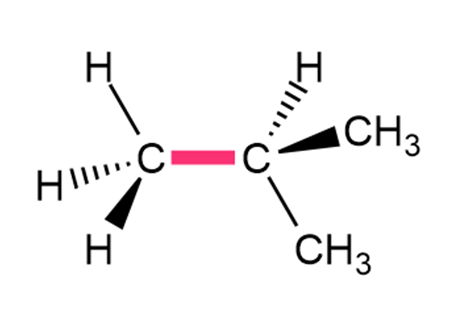
b.
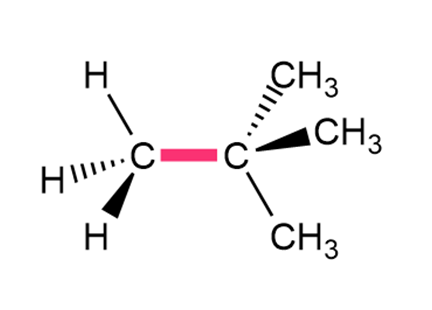
Short Answer
Answer
- The barrier to rotation is 16 kJ per mole.
- The barrier to rotation is 18 kJ per mole.
Learning Materials
Features
Discover
Chapter 4: Q.50. (page 128)
Question: Calculate the barrier to rotation for each bond highlighted in pink.
a.

b.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the five constitutional isomers having molecular formula .
Question: Convert each three-dimensional model to a Newman projection around the indicated bond.
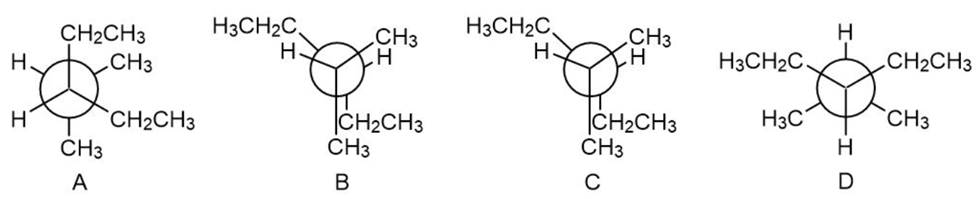
Question: Rank the following conformations in order of increasing energy.
Question: Which of the following is not another representation for 2-methylbutane?
a.
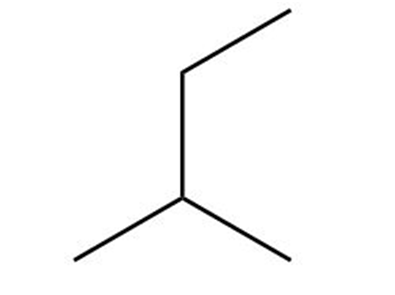
b.
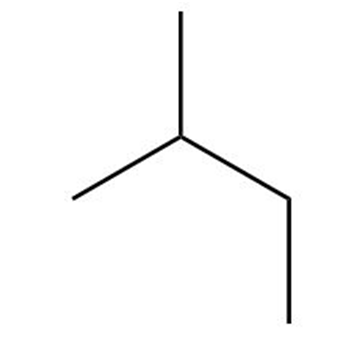
c.
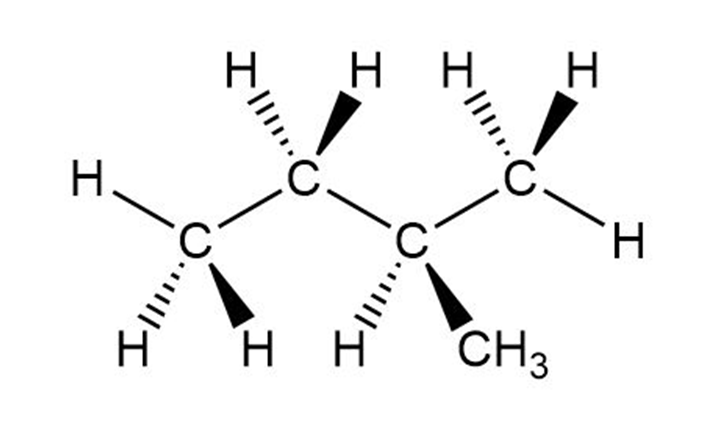
d.
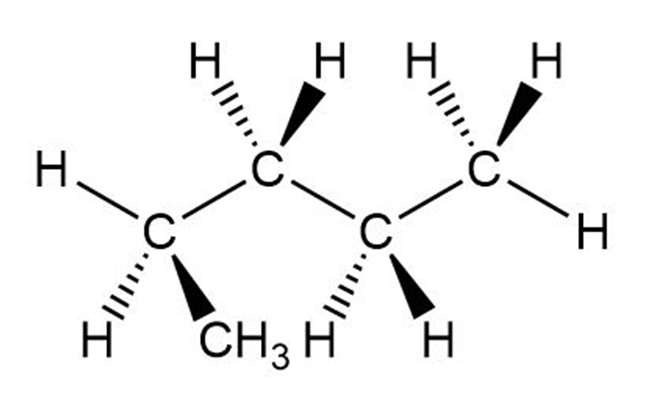
Question: Explain why beeswax is insoluble in , slightly soluble in ethanol , and soluble in chloroform role="math" localid="1648544714807" .
What do you think about this solution?
We value your feedback to improve our textbook solutions.