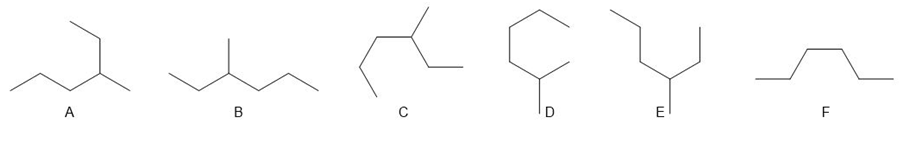
Chapter 4: Q.5. (page 128)
Question: a. Which compounds (B–F) are identical to A? b. Which compounds (B–F) represent an isomer of A?
Short Answer
Answer
a. B, C, and E are identical to A.
b. D and F are isomers of A.
Learning Materials
Features
Discover
Chapter 4: Q.5. (page 128)
Question: a. Which compounds (B–F) are identical to A? b. Which compounds (B–F) represent an isomer of A?

Answer
a. B, C, and E are identical to A.
b. D and F are isomers of A.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The eclipsed conformation of is role="math" localid="1648620042367" less stable than the staggered conformation. How much is the H, Cl eclipsing interaction worth in destabilization?
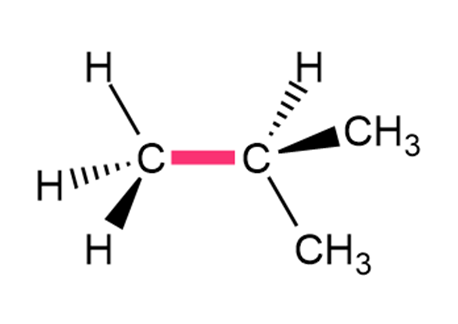
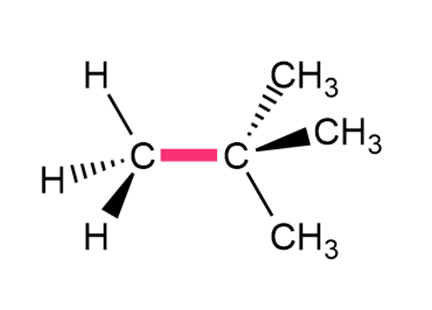
Question: Calculate the barrier to rotation for each bond highlighted in pink.
a.
b.
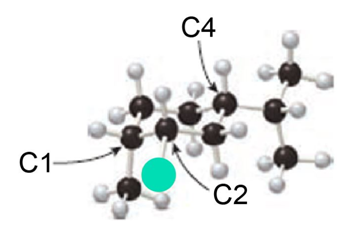
Question: Consider the substituted cyclohexane shown in the ball-and-stick model.
Question: Draw the products of combustion of each alkane.
a.
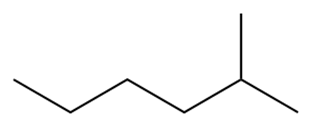
b.
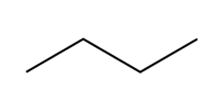
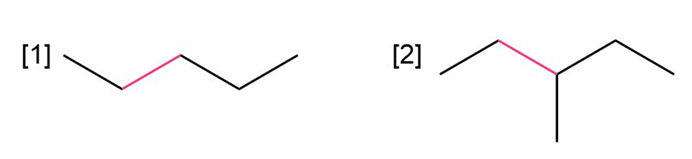
Question: a. Using Newman projections, draw all staggered and eclipsed conformations that result from rotation around the bond highlighted in pink in each molecule;
b. draw a graph of energy versus dihedral angle for rotation around this bond.
What do you think about this solution?
We value your feedback to improve our textbook solutions.