Chapter 4: Q.64. (page 128)
Question: Draw the products of combustion of each alkane.
a.
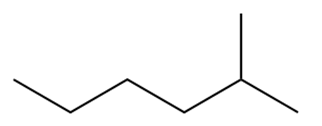
b.
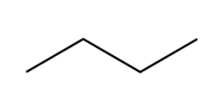
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 4: Q.64. (page 128)
Question: Draw the products of combustion of each alkane.
a.

b.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Haloethanes have similar barriers to rotation (13.4–15.5 kJ/mol) despite the fact that the size of the halogen increases, role="math" localid="1648639622891" . Offer an explanation.
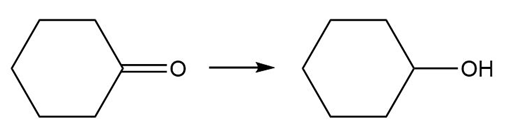
Question: Classify each transformation as an oxidation, reduction, or neither.
a.
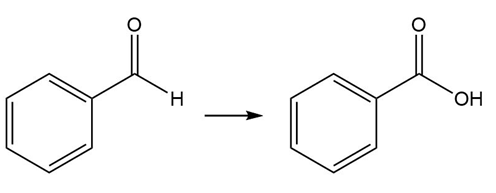
b.
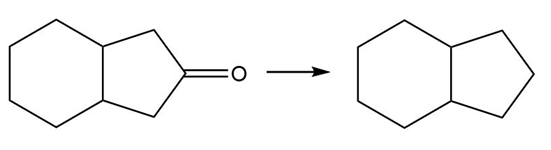
c.
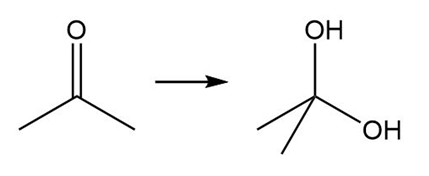
d.
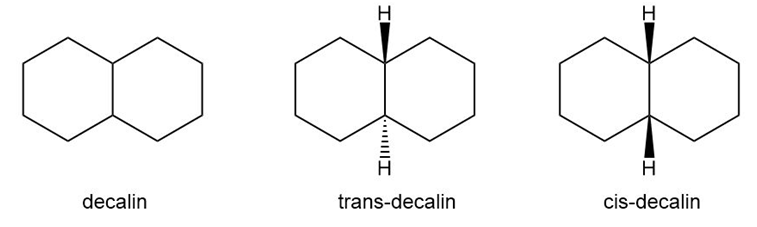
Question: When two six-membered rings share a C-C bond, this bicyclic system is called decalin. There are two possible arrangements: trans-decalin having two hydrogen atoms at the ring fusion on opposite sides of the rings, and cis-decalin having the two hydrogens at the ring fusion on the same side.
Give the structure corresponding to each IUPAC name.
a. 1,2-dimethylcyclobutane
b. 1,1,2-trimethylcyclopropane
c. 4-ethyl-1,2-dimethylcyclohexane
d. 1-sec-butyl-3-isopropylcyclopentane
e. 1,1,2,3,4-pentamethylcycloheptane
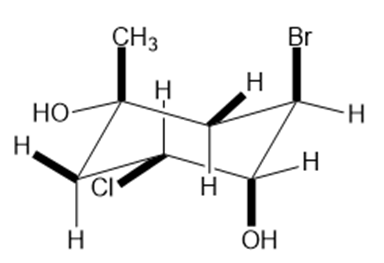
Question: Classify the ring carbons as up C’s or down C’s. Identify the bonds highlighted in bold as axial or equatorial.
What do you think about this solution?
We value your feedback to improve our textbook solutions.