Chapter 4: Q12. (page 140)
Give the structure corresponding to each IUPAC name.
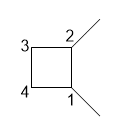
a. 1,2-dimethylcyclobutane
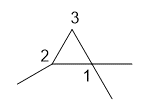
b. 1,1,2-trimethylcyclopropane
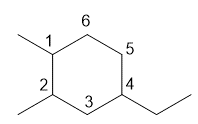
c. 4-ethyl-1,2-dimethylcyclohexane
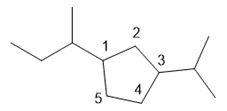
d. 1-sec-butyl-3-isopropylcyclopentane
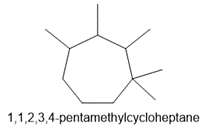
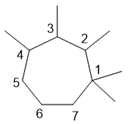
e. 1,1,2,3,4-pentamethylcycloheptane
Short Answer
a.
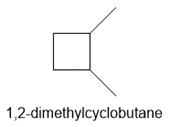
b.
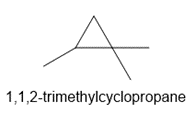
c.
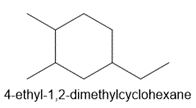
d.
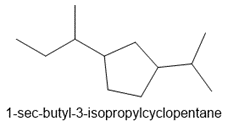
e.