Chapter 4: Q.13. (page 128)
Question: Arrange the following compounds in order of increasing boiling point.
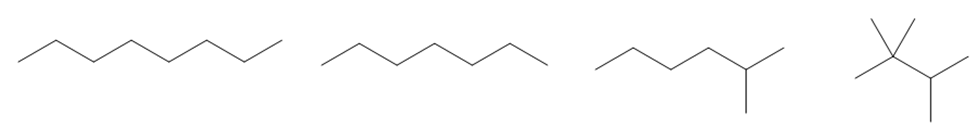
Short Answer
Answer
The order of increasing boiling point is given below:
Learning Materials
Features
Discover
Chapter 4: Q.13. (page 128)
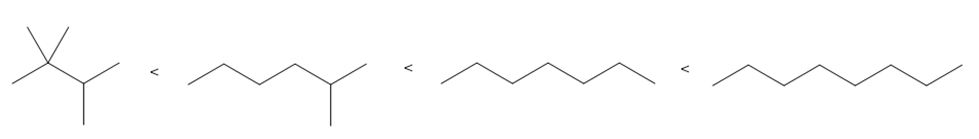
Question: Arrange the following compounds in order of increasing boiling point.

Answer
The order of increasing boiling point is given below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Each of the following IUPAC names is incorrect. Explain why it is incorrect and give the correct IUPAC name.
a. 2,2-dimethyl-4-ethylheptane
b. 5-ethyl-2-methylhexane
c. 2-methyl-2-isopropylheptane
d. 1,5-dimethylcyclohexane
e. 1-ethyl-2,6-dimethylcycloheptane
f. 5,5,6-trimethyloctane
g. 3-butyl-2,2-dimethylhexaneh. 1,3-dimethylbutane
Question: a. Draw the anti and gauche conformations for ethylene glycol .
b. Ethylene glycol is unusual in that the gauche conformation is more stable than the anti conformation. Offer an explanation.
Question: Hydrocarbons like benzene are metabolized in the body to arene oxides, which rearrange to form phenols. This is an example of a general process in the body, in which an unwanted compound (benzene) is converted to a more water-soluble derivative called a metabolite, so that it can be excreted more readily from the body.
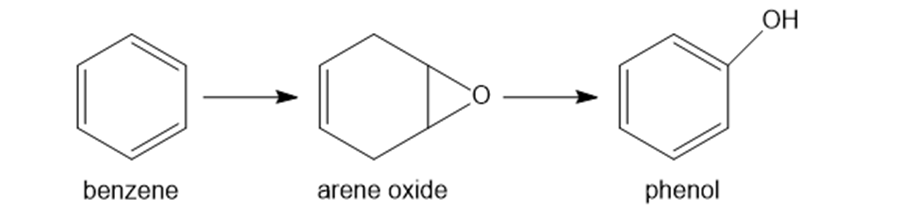
a. Classify each of these reactions as oxidation, reduction, or neither.
b. Explain why phenol is more water soluble than benzene. This means that phenol dissolves in urine, which is largely water, to a greater extent than benzene.
Question: For each compound drawn below:
a. Label each OH, Br, andgroup as axial or equatorial.
b. Classify each conformation as cis or trans.
c. Translate each structure into a representation with a hexagon for the six-membered ring, and wedges and dashed wedges for groups above and below the ring.
d. Draw the second possible chair conformation for each compound.
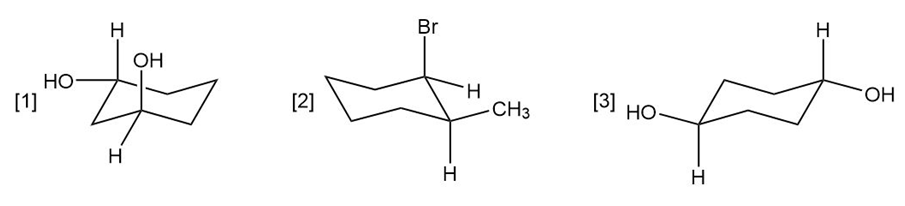
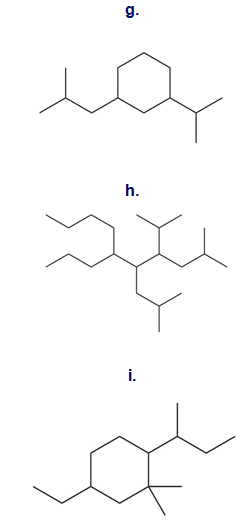
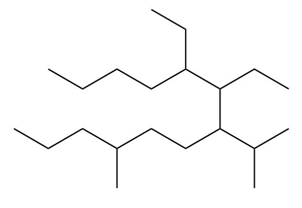
Question: Give the IUPAC name for each compound.
j
What do you think about this solution?
We value your feedback to improve our textbook solutions.