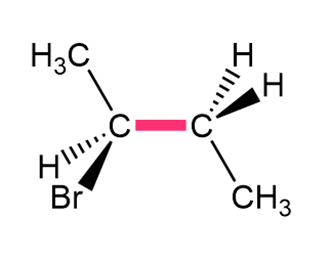
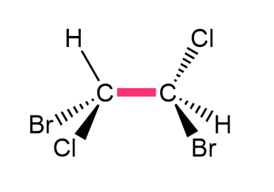
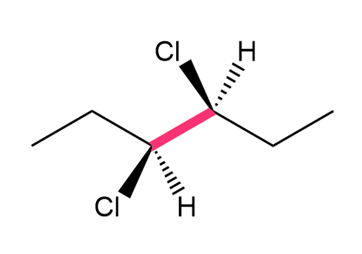
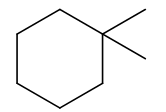
Chapter 4: Q.32. (page 128)
Question: Explain why beeswax is insoluble in , slightly soluble in ethanol , and soluble in chloroform role="math" localid="1648544714807" .
Short Answer
Answer
Beeswax is soluble in chloroform because it is the most non-polar solvent. It is insoluble in water because water is the most polar solvent.